Draw A Lewis Structure For Hcn
Draw A Lewis Structure For Hcn - Then, use single bonds to link carbon, hydrogen, and nitrogen. Figure out how many electrons the molecule must have, based on the number of valence electrons in each atom. Draw the lewis structure for hcn. Draw the lewis structure for the hcn molecule. Add these electrons to give every atom an octet. To draw the lewis structure of hcn, put carbon (c) in the middle, hydrogen (h) on one side, and nitrogen (n) on the other. #3 calculate and mark formal charges on the atoms, if required. Web drawing the lewis structure for hcn. Count the valence electrons you can use. Learning objectives in chapter 5. Draw the lewis structure for hcn. Count the valence electrons you can use. • how to draw lewis structure for pocl3. Include all lone pairs of electrons. As usual, this is the correct order of the atoms. We'll also compare hnc to hcn and discuss why both are of inter. As usual, this is the correct order of the atoms. To draw the lewis structure of hcn, put carbon (c) in the middle, hydrogen (h) on one side, and nitrogen (n) on the other. Web first, let's do hydrogen cyanide, the poison that might have killed lewis.. Web the lewis structure of hcn shows the arrangement of atoms and electrons in the molecule. Because of the 2 pi bonds and 1 sigma bond formed by the hybridization of 2px, 2py, and 2pz between c and n atoms, this 2p overlap makes the bond stronger and shorter therefore the bond between c and n is linear. Draw the. H + c + n =1 + 4 + 5 = 10. Count the valence electrons you can use. #3 calculate and mark formal charges on the atoms, if required. Web does this molecule | chegg.com. Be sure that you don't use more than the ten valence electrons available. To draw the lewis structure of hcn, put carbon (c) in the middle, hydrogen (h) on one side, and nitrogen (n) on the other. With the lewis structure for hcn you’ll need to share more than one pair of electrons between the carbon and the nitrogen atoms. The number of valence electrons in the molecule is (1 + 4 +. Put the least electronegative atom c in the middle with h and cl on either side. To learn the lewis acid structure, molecular mass, physical and chemical properties and uses with faqs of hydrogen cyanide visit byju’s. Does this compound have any plausible resonance structures? #2 mark lone pairs on the atoms. (note that we denote ions with brackets around. You nave to put a triple bond between c and n. #2 mark lone pairs on the atoms. As usual, this is the correct order of the atoms. To draw the lewis structure of hcn, put carbon (c) in the middle, hydrogen (h) on one side, and nitrogen (n) on the other. Use these steps to correctly draw the hcn. Web drawing lewis structures for molecules with one central atom: Learning objectives in chapter 5. The number of valence electrons in the molecule is (1 + 4 + 5) = 10. Web lewis dot structure can be used to get the basic idea of the structure. Figure out how many electrons the molecule must have, based on the number of. When i'm putting the electrons in, i usually start by putting each atom's valence electrons around it, then i connect the dots into. Draw the lewis structure for hcn. The hcn molecule consists of three atoms: Be sure that you don't use more than the ten valence electrons available. Count the valence electrons you can use. Web drawing lewis structures for molecules with one central atom: Use these steps to correctly draw the hcn lewis structure: Draw the lewis structure for the hcn molecule. H + c + n =1 + 4 + 5 = 10. #2 mark lone pairs on the atoms. Draw the lewis structure for the hcn molecule. With the lewis structure for hcn you’ll need to share more than one pair of electrons between the carbon and the nitrogen atoms. Learning objectives in chapter 5. Hydrogen (h), carbon (c), and nitrogen (n). Here's how to do it. Put the least electronegative atom c in the middle with h and cl on either side. The number of valence electrons in the molecule is (1 + 4 + 5) = 10. Web chemistry questions and answers. Use these steps to correctly draw the hcn lewis structure: Web how to draw the lewis structure of hcn? As usual, this is the correct order of the atoms. Figure out how many electrons the molecule must have, based on the number of valence electrons in each atom. You nave to put a triple bond between c and n. Be sure that you don't use more than the ten valence electrons available. 4k views 6 years ago chem 101: To learn the lewis acid structure, molecular mass, physical and chemical properties and uses with faqs of hydrogen cyanide visit byju’s.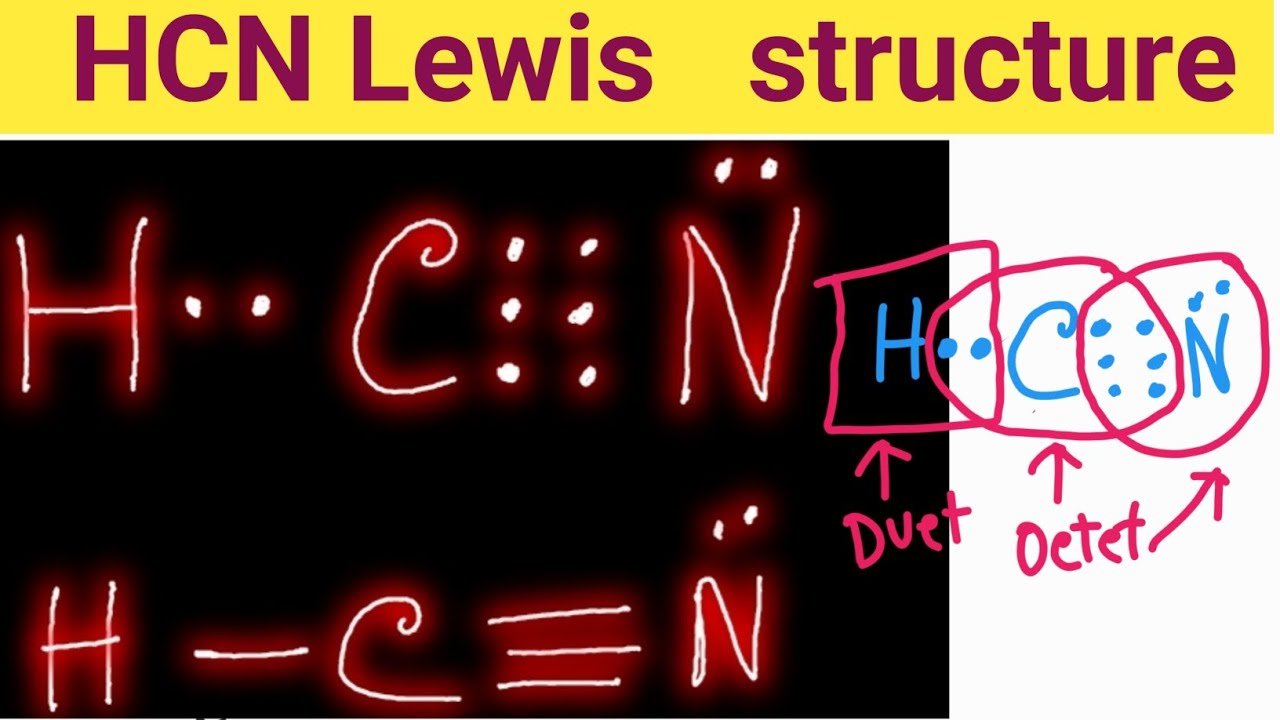
HCN Lewis StructureHydrogen Cyanide (HCN) Lewis Dot StructureDraw

Draw the Lewis dot structure of Hydrogen cyanide (HCN) molecule

Lewis structure of HCN (Hydrogen cyanide) YouTube

Lewis Diagram For Hcn
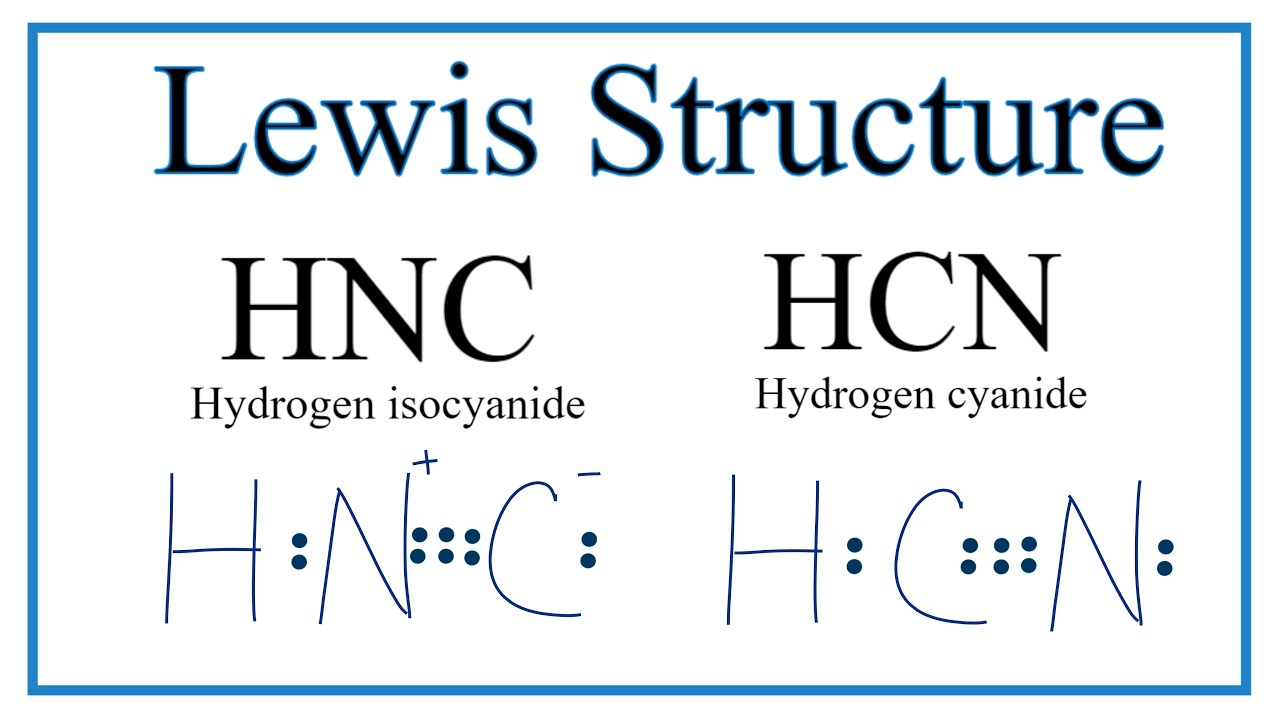
How to Draw the Lewis Dot Structure for HNC Hydrogen Isocyanide YouTube

Estrutura De Lewis Hcn
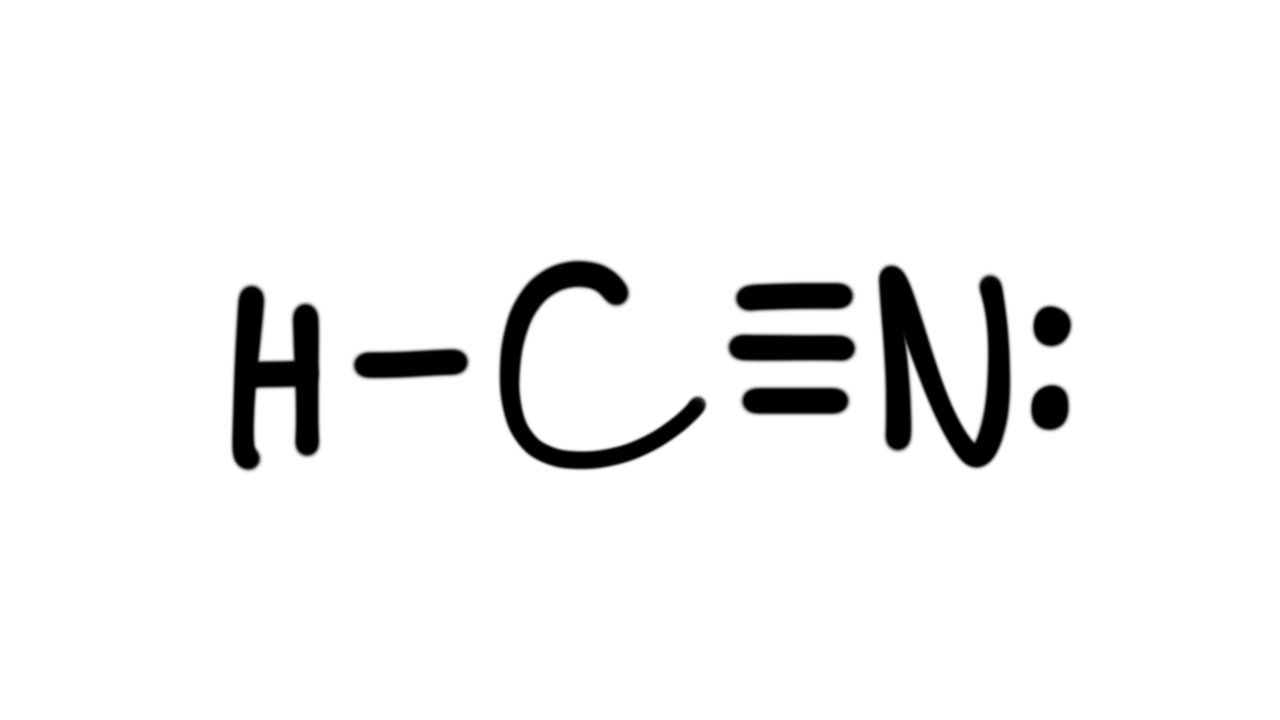
Estrutura De Lewis Hcn

How to Draw the Lewis Diagram for HCN StepbyStep Guide

Hcn hydrogen cyanide molecule Royalty Free Vector Image
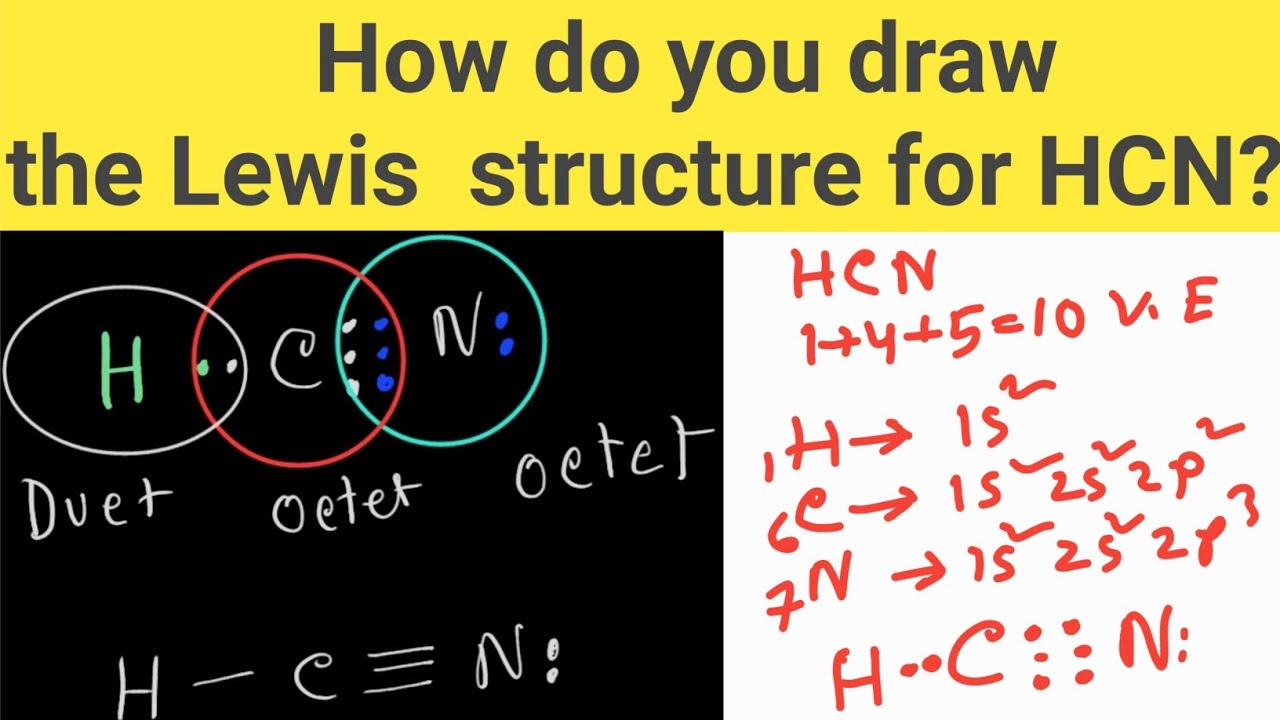
How do you draw the Lewis structure of HCN (hydrogen cyanide)? HCN
Add These Electrons To Give Every Atom An Octet.
The Lewis Structure Of Hcn Shows That The Carbon Atom Is The Central Atom And Is Covalently Bonded To Both Hydrogen And Nitrogen Atoms.
#1 First Draw A Rough Sketch.
We'll Also Compare Hnc To Hcn And Discuss Why Both Are Of Inter.
Related Post: