Draw Lewis Structure For H2S
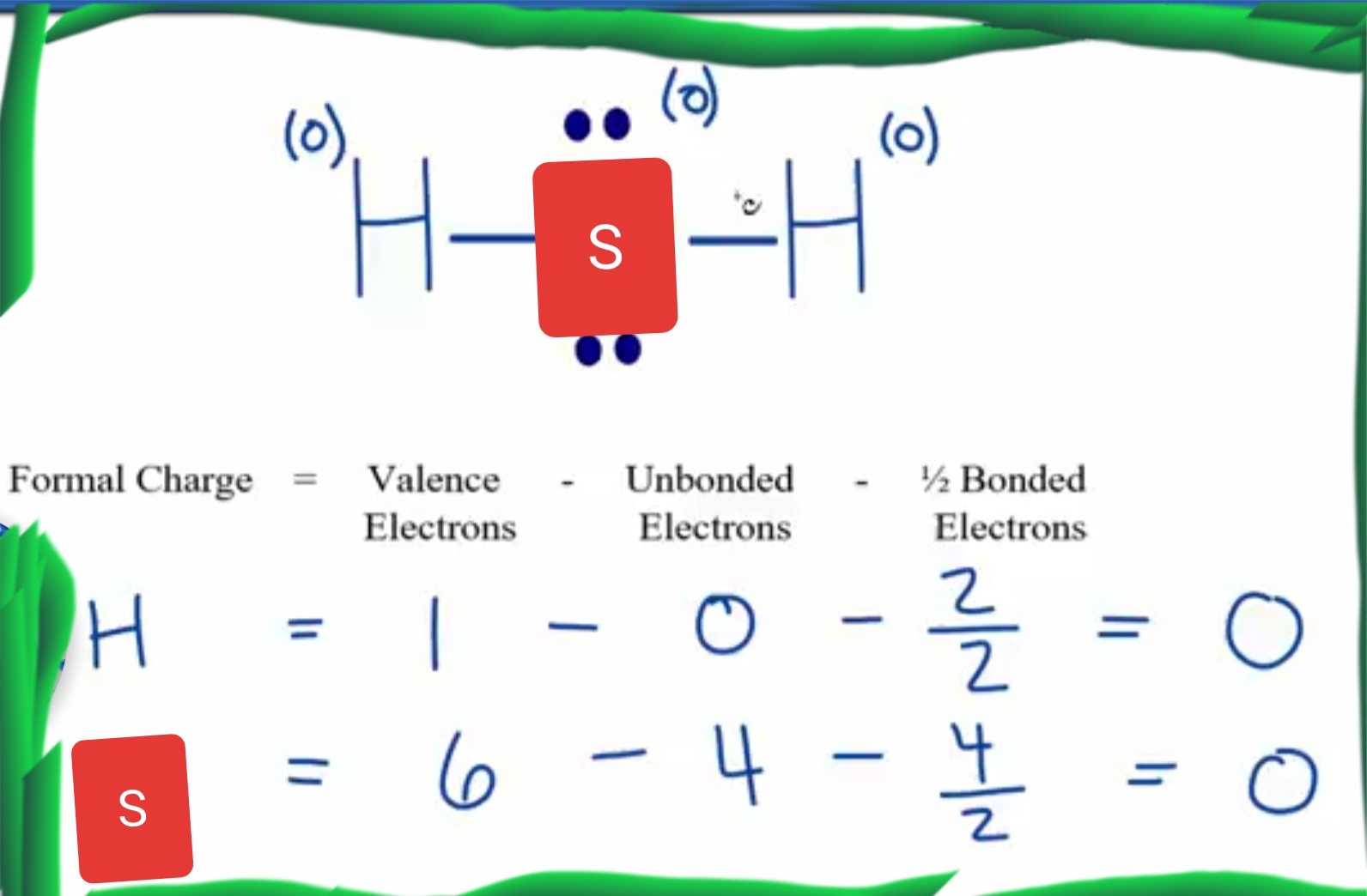
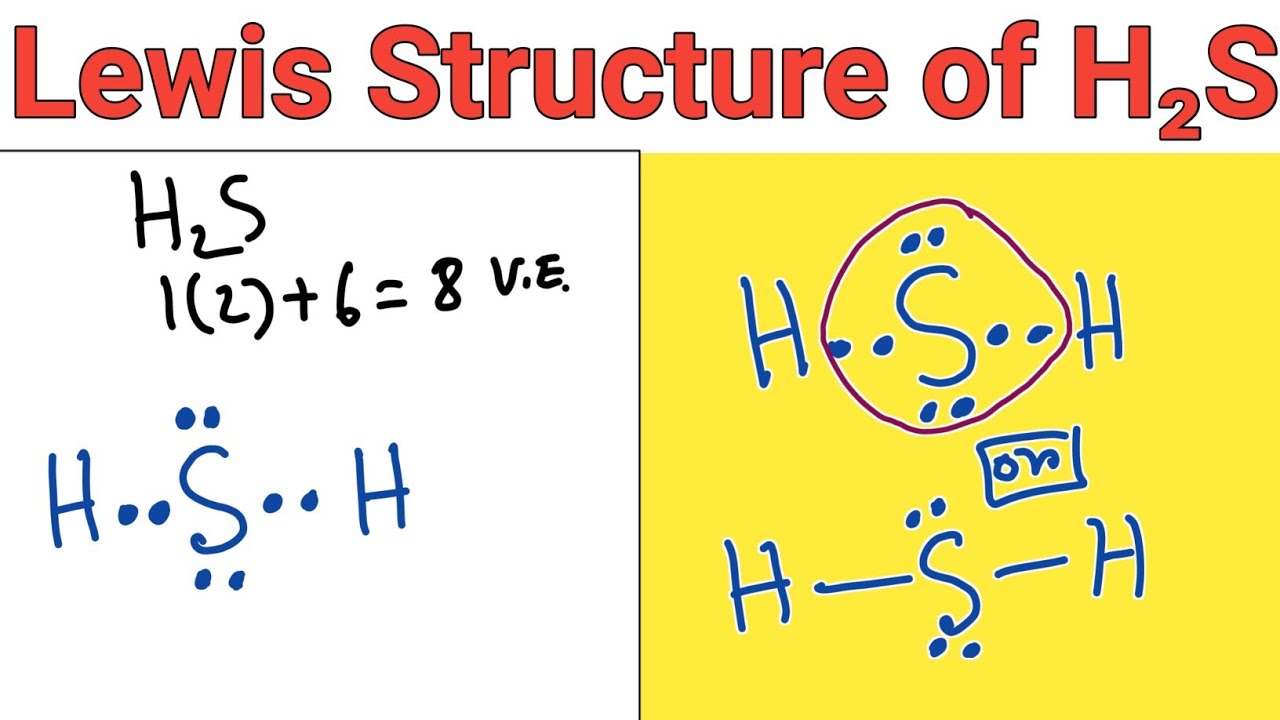
Draw Lewis Structure For H2S - Let’s break down each step in more detail. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. To accurately represent the h2s lewis structure, we need to calculate the total valence electrons. Web drawing lewis structures for molecules with one central atom: 2 electrons (1 electron per atom) x 2 atoms = 2 electrons First, determine the total number of valence electrons. Breaking the octet rule ; In this compound valence electron will be as following: Draw the electron dot stucture of f 2 (fluroine). How to draw the dot structure for h2s; Sulfur needs eight electrons to fulfill the requirements for octet rule. Find more chemistry widgets in wolfram|alpha. Web what is the lewis structure of hydrogen sulfide h2s? Hydrogen has one valence electron, while sulfur has six valence electrons. For the h2s structure use the periodic table to find the total number of valence electrons for. There are 2 lone pairs on the sulfur atom (s). How to draw the dot structure for h2s; Web get the free lewis structure finder widget for your website, blog, wordpress, blogger, or igoogle. Breaking the octet rule ; Count the total number of valence electrons; You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Web how do you arrange the electrons around the atoms of hydrogen sulphide? Valence electrons in hydrogen (h): Web draw the skeletal of h2s lewis structure: Web drawing lewis structures for molecules with one central atom: Draw the lewis structure of hydrogen sulfide (h₂s) and then determine its electron domain and molecular geometries. Valence electrons in hydrogen (h): Sulfur needs eight electrons to fulfill the requirements for octet rule. Determining the total valence electrons. Calculate the total number of valence electrons. Using the periodic table to draw lewis dot structures. Sum the valence electrons of each atom: Web let's do the lewis structure for h2s: Watch this video to learn how to draw the lewis dot structure of h2s, a simple but important molecule in chemistry and biology. In this compound, both the hydrogen atoms require one electron to make the. Using the periodic table to draw lewis dot structures. #4 calculate formal charge and check stability. This h2s lewis structure follows the octet rule with electron sharing and also has zero formal charge. Calculate the total number of valence electrons. Valence electrons in hydrogen (h): Breaking the octet rule ; Web what is the lewis structure of hydrogen sulfide h2s? Play quiz game > +1 vote. Examples for drawing lewis structures for covalent bonds. First, determine the total number of valence electrons. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. Check the octet rule (formal charges) step 6: Count the total number of valence electrons; Now to understand this we need to know the steps to draw a lewis structure at first. Web get the free lewis structure finder widget for your website, blog,. Using the periodic table to draw lewis dot structures. Now to understand this we need to know the steps to draw a lewis structure at first. Web how do you arrange the electrons around the atoms of hydrogen sulphide? #1 draw a rough sketch of the structure. Using formal charges to determine how many bonds to make, a different perspective. Plus sulfur is in group 6 or 16 on the periodic table, so it has 6 valence electrons. Find the total number of valence electrons of hydrogen and sulfur atoms. Examples for drawing lewis structures for covalent bonds. In this compound valence electron will be as following: Draw the electron dot structure for h2s. As hydrogen atom can’t coordinate with more than one atom so sulfur become central atom and two hydrogen atoms are written on the opposite sides. Web learn the steps to draw the lewis structure of h2s (dihydrogen sulfide) in just 1 minute.📌you can draw any lewis structures by following the simple steps me. Sulfur needs eight electrons to fulfill the requirements for octet rule. Breaking the octet rule ; Plus sulfur is in group 6 or 16 on the periodic table, so it has 6 valence electrons. Web the albert team. Hydrogen has one valence electron, while sulfur has six valence electrons. Answered jan 24, 2020 by surajkumar (64.8k points) selected jan 25, 2020 by nishu03. #1 draw a rough sketch of the structure. Web draw the skeletal of h2s lewis structure: Find the total number of valence electrons of hydrogen and sulfur atoms. #1 draw a rough sketch of the structure. Draw the lewis structure of hydrogen sulfide (h₂s) and then determine its electron domain and molecular geometries. 2 electrons (1 electron per atom) x 2 atoms = 2 electrons #4 calculate formal charge and check stability. Here, the given molecule is h2s (dihydrogen sulfide).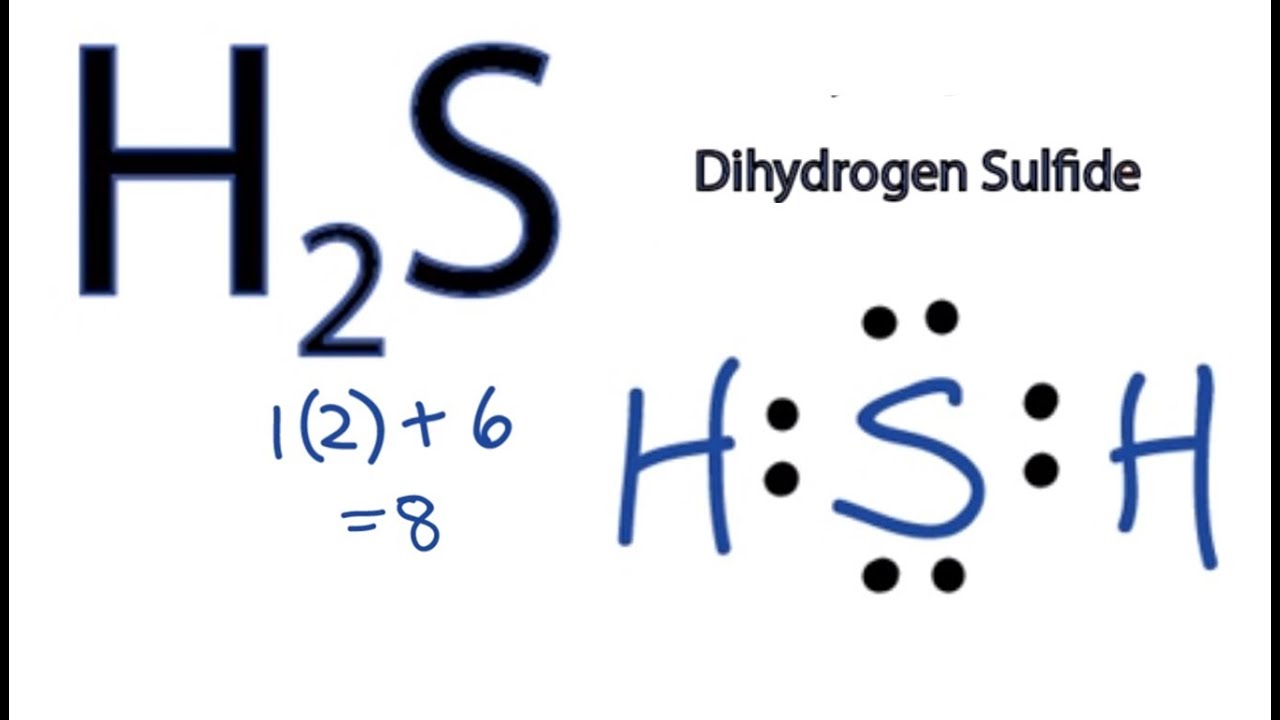
H2S Lewis Structure How to Draw the Dot Structure for H2S YouTube

H2S Lewis Structure, Molecular Geometry, Hybridization, and MO Diagram
【4 Steps】H2S Lewis StructureLewis Structure for H2S (Dihydrogen

H2S Lewis Structure ,Valence Electrons ,Formal Charge,Polar or Nonpolar
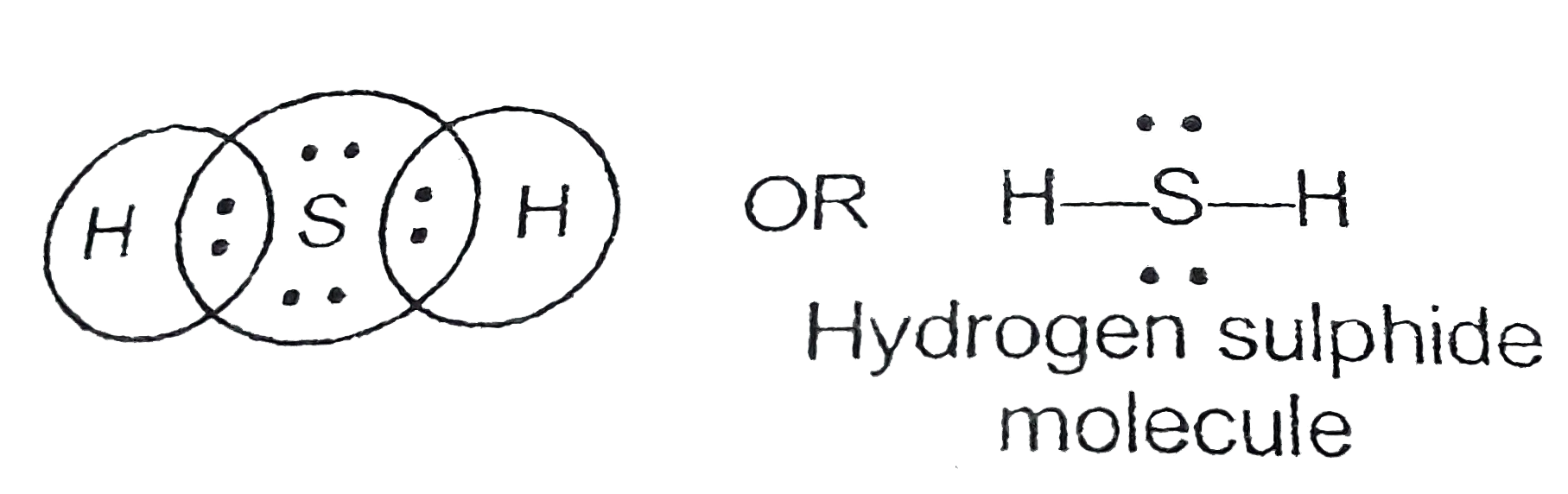
31+ H2S Lewis Structure Pictures Bepe Enthusiastic

Lewis Structure Hydrogen Sulfide H2s Stock Vector (Royalty Free
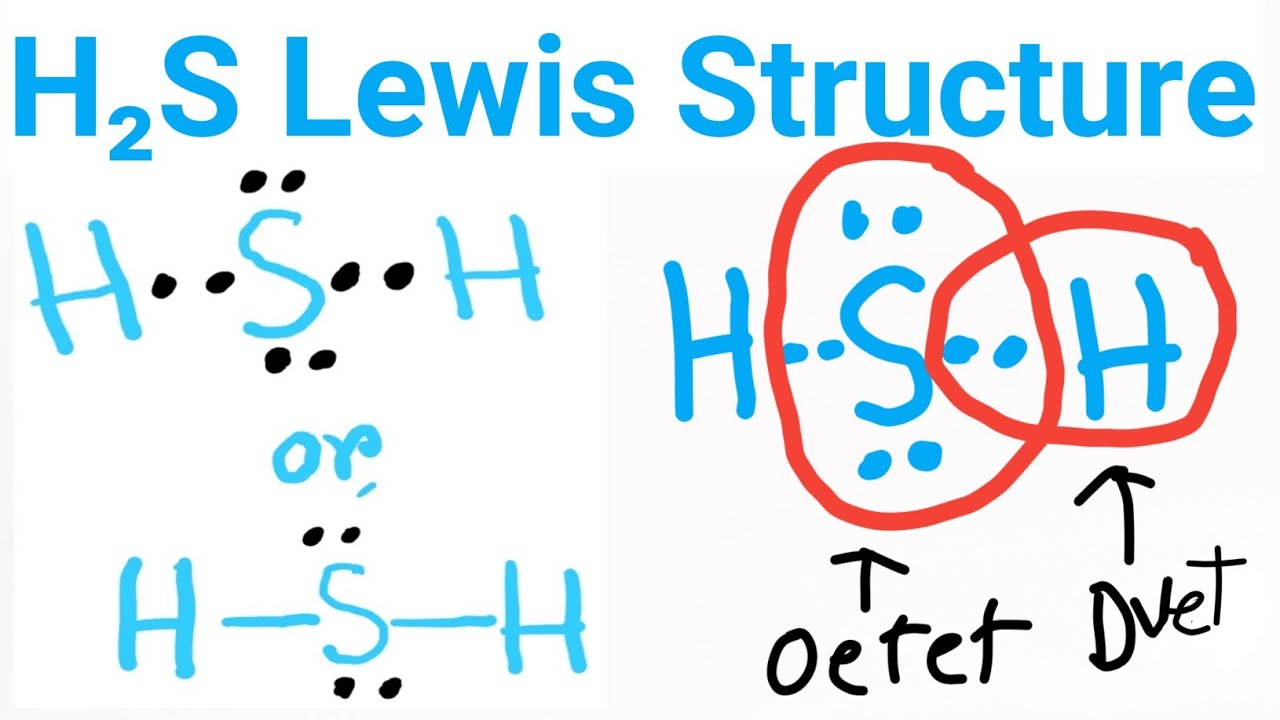
H2S Lewis Structure Lewis Dot Structure for H2S Hydrogen sulfide
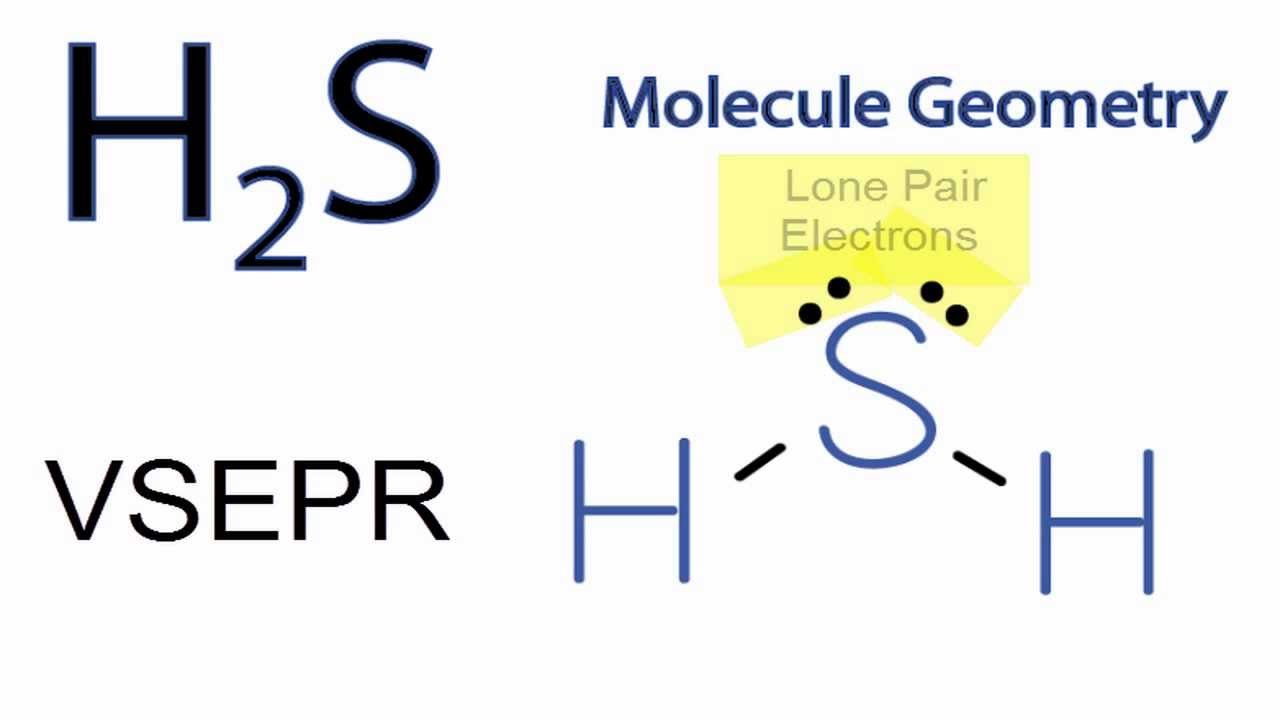
H2S Lewis Structure, Molecular Geometry, Hybridization and Polarity
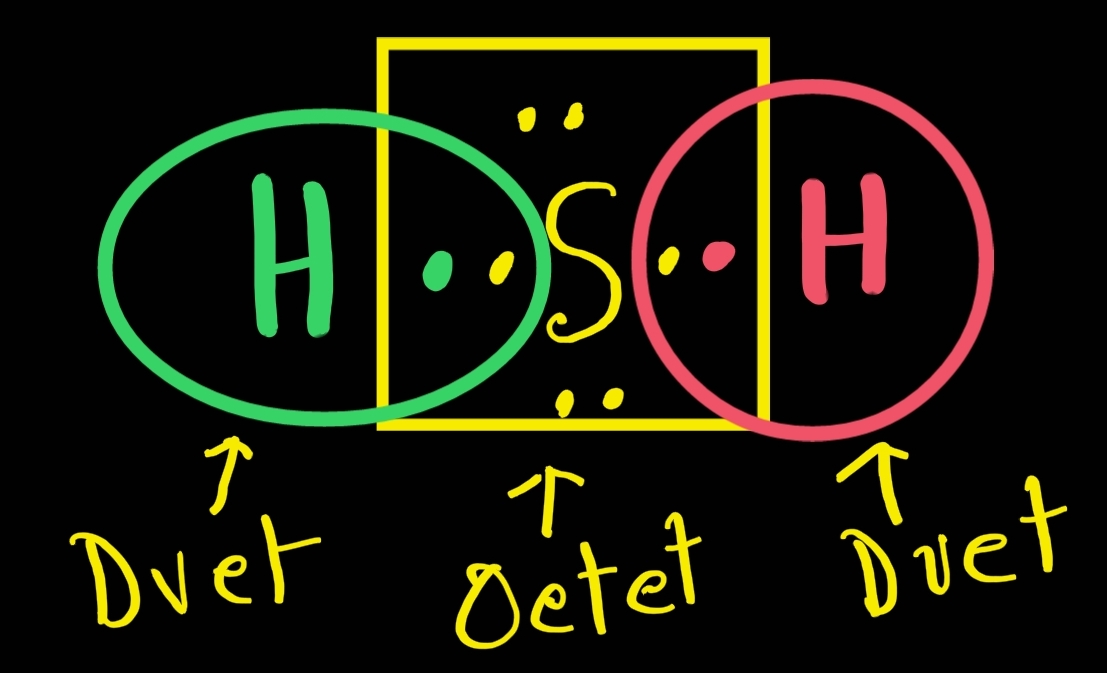
H2S Lewis Structure ,Valence Electrons ,Formal Charge,Polar or Nonpolar
Lewis structure of H2S (Hydrogen sulphide) YouTube
Using The Periodic Table To Draw Lewis Dot Structures.
#3 Indicate Formal Charges On The Atoms, If Necessary.
Hydrogen, Group 1, Has 1 Valence Electron, But We Have Two Hydrogens Here So Let's Multiply That By 2.
Web What Is The Lewis Structure Of Hydrogen Sulfide H2S?
Related Post: