Draw The Lewis Structure For A Sulfur Monoxide Molecule
Draw The Lewis Structure For A Sulfur Monoxide Molecule - Do not add any more atoms. Web to draw the lewis structure for so (sulfur monoxide), we need to consider the valence electrons of sulfur and oxygen. Sulfur is in group 6a and has 6 valence electrons, while oxygen is in group 6a and also has 6 valence electrons. Web the lewis structure for disulfur monoxide is shown below. Web we can draw the lewis structure of any covalent molecule by following the six steps discussed earlier. Web an example of a stable molecule with an odd number of valence electrons would be nitrogen monoxide. For very simple molecules and molecular ions, we can write the lewis structures by merely pairing up the unpaired electrons on the. Explain the concept of resonance and draw lewis structures representing resonance. S •••••• o so, the lewis structure for sulfur. See examples of lewis structures for. Web draw lewis structures depicting the bonding in simple molecules. The s−o bond length of 148.1 pm is. In this case, we can condense the last few steps, since not all of. Sulfur is in group 6a and has 6 valence electrons, while oxygen is in group 6a and also has 6 valence electrons. Web learn how to write lewis. To find the lewis structure of sulfur monoxide, enter so or s=o in the input box and click get the lewis structure. Find out the valence electrons, the center atom, the chemical bond, the octet rule, and the formal. Web drawing lewis structures for molecules with one central atom: In total, we have 12 valence. Web the lewis structure for. Web complete the lewis structures of these molecules by adding multiple bonds and lone pairs. In total, we have 12 valence. Submitted by robert v., oct. We can draw the lewis structure of any covalent molecule by following the six steps discussed earlier. Web this widget gets the lewis structure of chemical compounds. Draw the lewis structure for sulfur monoxide (so) molecule. Sulfur has 6 valence electrons, and oxygen has 6 valence electrons. We can draw the lewis structure of any covalent molecule by following the six steps discussed earlier. Starting from this structure, complete the lewis structure that follows the octet rule on all atoms. Web the lewis structure for disulfur monoxide. Do not add any more atoms. See the full solution and post any questions on chegg.com. We also use lewis symbols to indicate the formation of covalent bonds, which are shown in. We can draw the lewis structure of any covalent molecule by following the six steps discussed earlier. Find out the valence electrons, lone pairs, formal charges and. Web learn how to draw the lewis structure of s2o (disulfur monoxide) with 6 simple steps and images. We also use lewis symbols to indicate the formation of covalent bonds, which are shown in. Web to draw the lewis structure for so (sulfur monoxide), we need to consider the valence electrons of sulfur and oxygen. Explain the concept of resonance. Web an example of a stable molecule with an odd number of valence electrons would be nitrogen monoxide. Submitted by robert v., oct. The following procedure will give you the correct lewis structure for any molecule or. Web explain the concept of resonance and draw lewis structures representing resonance forms for a given molecule; Find out the valence electrons, center. Calculate the formal charge for each atom in the carbon. Do not add any more atoms. Sulfur has 6 valence electrons, and oxygen has 6 valence electrons. Determine the total number of valence electrons in sulfur monoxide. Web the lewis structure for ammonia (nh₃) shown below is incorrect. Web draw lewis structures for molecules. Web an example of a stable molecule with an odd number of valence electrons would be nitrogen monoxide. Find out the valence electrons, the center atom, the chemical bond, the octet rule, and the formal. Web drawing lewis structures for molecules with one central atom: Sulfur has 6 valence electrons, and oxygen has 6. Web draw lewis structures depicting the bonding in simple molecules. Web the lewis structure for ammonia (nh₃) shown below is incorrect. Calculate the formal charge for each atom in the carbon. Web learn how to draw the stable lewis structure of so, a sulfur monoxide molecule, with six steps and examples. Web drawing lewis structures for molecules with one central. Web we can draw the lewis structure of any covalent molecule by following the six steps discussed earlier. Do not add any more atoms. Web draw lewis structures depicting the bonding in simple molecules. Therefore, the total number of. Web in this case, both sulfur and oxygen have satisfied the octet rule. The lewis structure for sulfur monoxide (so) is as follows: Web an example of a stable molecule with an odd number of valence electrons would be nitrogen monoxide. Web this widget gets the lewis structure of chemical compounds. Submitted by robert v., oct. Find out the valence electrons, lone pairs, formal charges and. In this case, we can condense the last few steps, since not all of them. This web page does not cover sulfur monoxide or how. Explain the concept of resonance and draw lewis structures representing resonance. Web learn how to draw the lewis dot structure of so with 6 simple steps and images. In total, we have 12 valence. Web to draw the lewis structure for so (sulfur monoxide), we need to consider the valence electrons of sulfur and oxygen.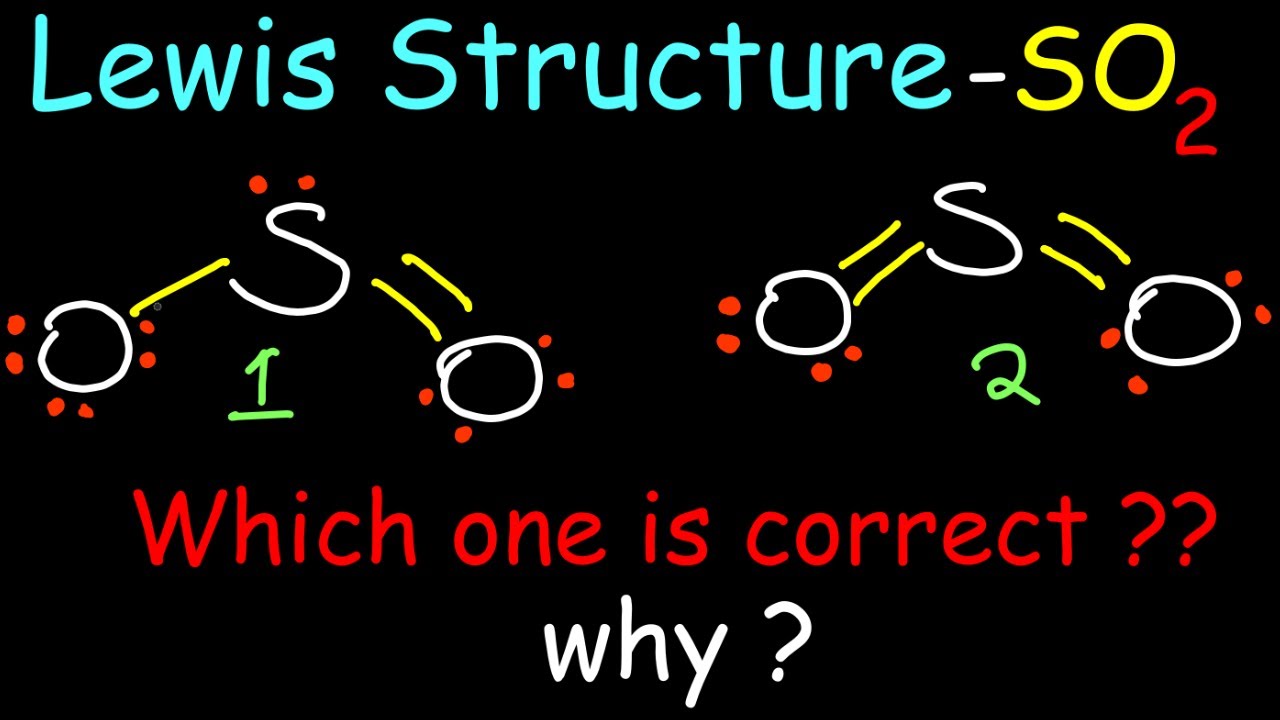
Lewis Structure of Sulphur Dioxide SO2 YouTube

So sulfur monoxide molecule Royalty Free Vector Image
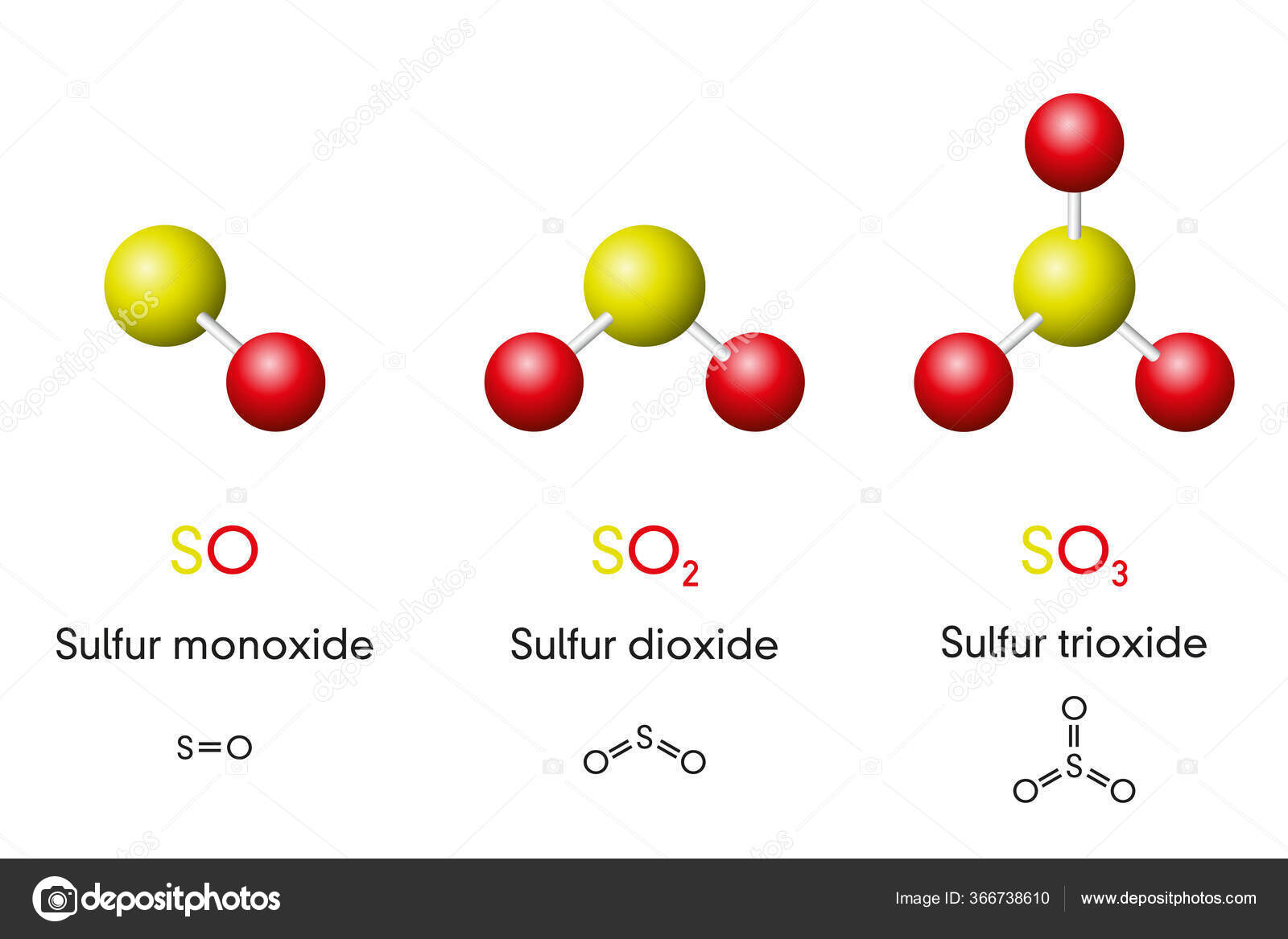
Three Sulfur Oxides Molecule Models Chemical Formulas Sulfur Monoxide

How to Write the Formula for Sulfur monoxide YouTube
Draw the Lewis Structure for a Sulfur Monoxide So Molecule

36 lewis dot diagram for sulfur Diagram Resource 2022

Draw the Lewis Structure for a Sulfur Monoxide So Molecule
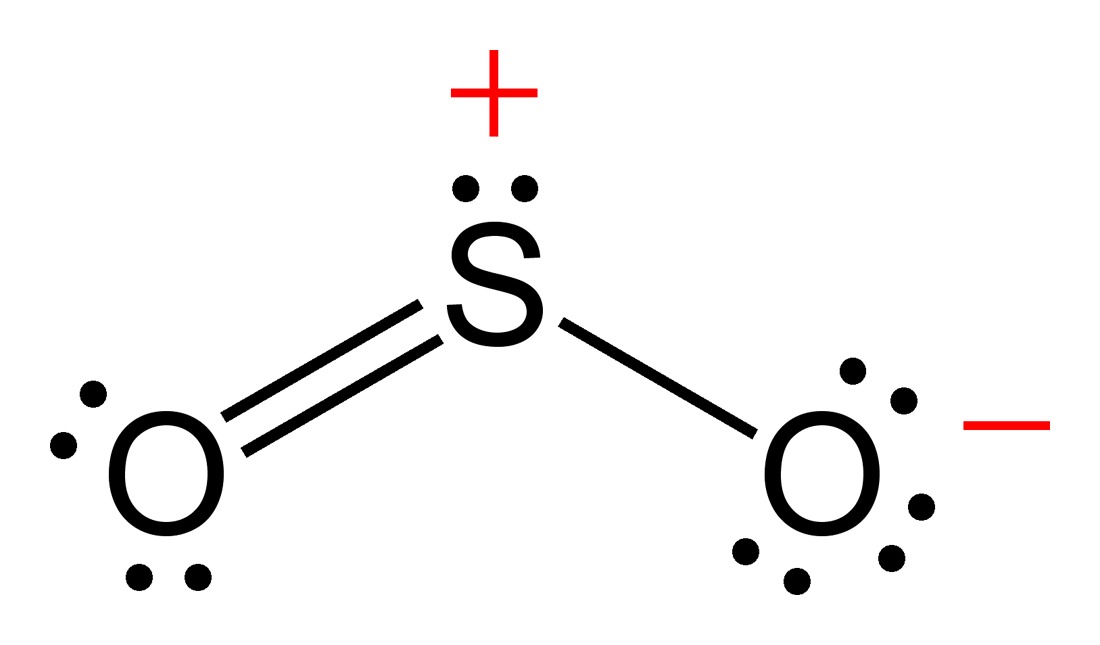
SO2(Sulfur Dioxide) Molecular Geometry & Lewis Structure Geometry of

SOLVED Draw the Lewis structure for sulfur monoxide (SO) molecule

Draw the Lewis Structure for a Sulfur Monoxide So Molecule
Find Out The Valence Electrons, Center Atom, Bonds, Octet Rule, And Formal.
The S−O Bond Length Of 148.1 Pm Is.
Web Learn How To Calculate Formal Charges And Use Them To Identify The Most Reasonable Lewis Structure For A Given Molecule.
Web Complete The Lewis Structures Of These Molecules By Adding Multiple Bonds And Lone Pairs.
Related Post: