Draw The Lewis Structure For Cocl2
Draw The Lewis Structure For Cocl2 - One uses math, the other puzzle pieces to give the three correct structure. A lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. You'll get a detailed solution from a subject matter expert that helps you learn core concepts. 1×4 = 4 cl : 4 + (3 × 6) + 2 = 24 electrons. Therefore, the lewis structure for the cocl 2 is represented as follows:. Draw the lewis structure for cocl2, including lone pairs. Alternatively a dot method can be used to draw the lewis structure. Web steps of drawing cocl2 lewis structure step 1: Each hydrogen atom (group 1) has one valence electron, carbon (group 14) has 4 valence electrons, and oxygen (group 16) has 6 valence. The lewis structure is the representation of valence electrons in a molecule. Because carbon is less electronegative than oxygen and hydrogen is normally terminal, c must be the central atom. Find more chemistry widgets in wolfram|alpha. The final answer must have this number of electrons‼! 8 + (2 × 7) = 22. Shared pairs of electrons are drawn as lines between atoms, while lone pairs of electrons are drawn as dots next to atoms. Alternatively a dot method can be used to draw the lewis structure. Web steps of drawing cocl2 lewis structure step 1: What is the molecular shape of cocl2 ? (valence electrons are the electrons that are present in. Web 6 steps to draw the lewis structure of cocl2 step #1: Web this problem has been solved! Include all lone pairs of electrons. Web the formal charges being 0 for all of the atoms in the cocl 2 molecule tells us that the lewis dot structure presented above is stable. 8 + (6 × 7) = 50. We follow the following steps to draw the lewis structure of the molecule. Web hello guys!phosgene having a chemical formula of cocl2 is a polyatomic molecule. Web drawing the lewis structure for cocl 2. Bent linear tetrahedral trigonal planar trigonal pyramidal what is the cl−c−cl bood angle?what is the molecular shape of cocl2 ? Alternatively a dot method can be. Web the formal charges being 0 for all of the atoms in the cocl 2 molecule tells us that the lewis dot structure presented above is stable. Draw the lewis structure of phosgene, cocl2. Calculate the total number of valence electrons. Web we can draw the lewis structure of any covalent molecule by following the six steps discussed earlier. (valence. Alternatively a dot method can be used to draw the lewis structure. Because carbon is less electronegative than oxygen and hydrogen is normally terminal, c must be the central atom. Web the ch2o c h 2 o molecule. Web this problem has been solved! Web we show two methods to find correct lewis structure of cocl2. Web to properly draw the cocl 2 lewis structure, follow these steps: (assign lone pairs, radical electrons, and atomic charges where appropriate.) calculate the electrons required (er), valence electrons (ve), and shared pairs (sp). Here, the given molecule is cocl2. Step 2) attach the atoms to each other using single bonds (“draw the skeleton structure”) step 3) add electrons to. For cocl 2, it is as shown below: In this case, we can condense the last few steps, since not all of them apply. In order to find the total valence electrons in a cocl2 molecule, first of all you should know the valence electrons present in carbon atom, oxygen atom as well as chlorine atom. In the lewis structure. All atoms have formal charges equals to 0 in this structure. 8 + (2 × 7) = 22. Enter the lewis structure for the molecule of cocl2. For cocl 2, it is as shown below: Web get the free lewis structure finder widget for your website, blog, wordpress, blogger, or igoogle. 1×4 = 4 cl : One uses math, the other puzzle pieces to give the three correct structure. Web the formal charges being 0 for all of the atoms in the cocl 2 molecule tells us that the lewis dot structure presented above is stable. Determine the lewis structure of the molecule. Enter the lewis structure for the molecule of. Find the total valence electrons in cocl2 molecule. It has a boiling point (b.p.) of around 8.3 0c. To determine the hybridization of cobalt. Find more chemistry widgets in wolfram|alpha. Phosgene is a colorless gaseous compound known as carbonyl chloride and has a molecular weight of 98.92 gram/mol. Web get the free lewis structure finder widget for your website, blog, wordpress, blogger, or igoogle. #1 draw a rough sketch of the structure #2 next, indicate lone pairs on the atoms #3 indicate formal charges on the atoms, if necessary #4 minimize formal charges by converting lone pairs of the atoms #5 repeat step 4 if necessary, until all charges are minimized When constructing a lewis diagram, keep in mind the octet rule, which refers to the tendency. Bent linear tetrahedral trigonal planar trigonal pyramidal what is the cl−c−cl bond angle? Therefore, the lewis structure for the cocl 2 is represented as follows:. It is used widely to make plastics and pesticides. Web this is the complete lewis structure of co 2. Calculate the total valence electrons in the molecule. Calculate the number of valence electrons: Web we show two methods to find correct lewis structure of cocl2. Step 2) attach the atoms to each other using single bonds (“draw the skeleton structure”) step 3) add electrons to all outer atoms (except h) to complete their octets.
COCl2 Lewis Structure (Phosgene) YouTube

COCl2 Lewis Structure How to Draw the Lewis Structure for COCl2 YouTube

COCl2 Lewis Structure Hybridization Polarity Molecular Geometry

Lewis Dot Structure For Cocl2

Cocl2 Lewis Structure Shape Draw Easy
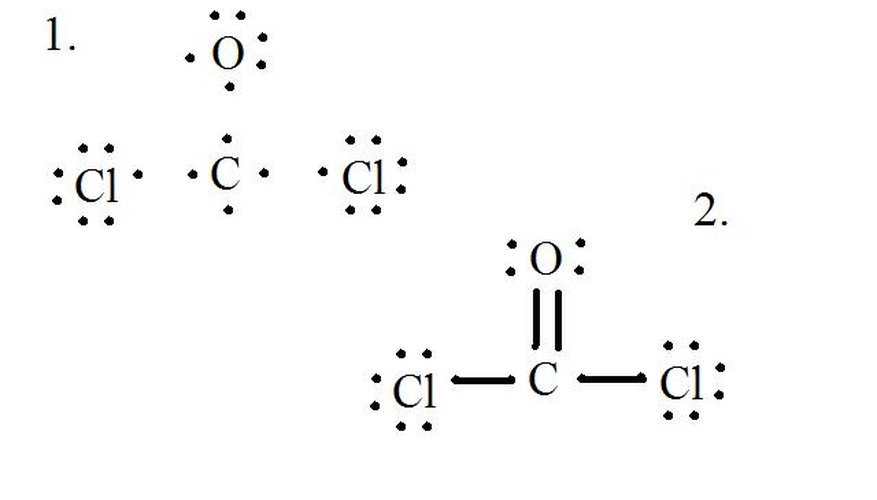
How to Calculate the Formal Charge of CoCl2 Sciencing
![The Lewis Structure of COCl2 [with free study guide and video]](https://www.aceorganicchem.com/blog/wp-content/uploads/2023/05/COCl2-lewis-structure.jpg)
The Lewis Structure of COCl2 [with free study guide and video]

Cocl2 Lewis Dot Structure Draw Easy
SOLVED Draw the Lewis structure for COCl2, including lone pairs.
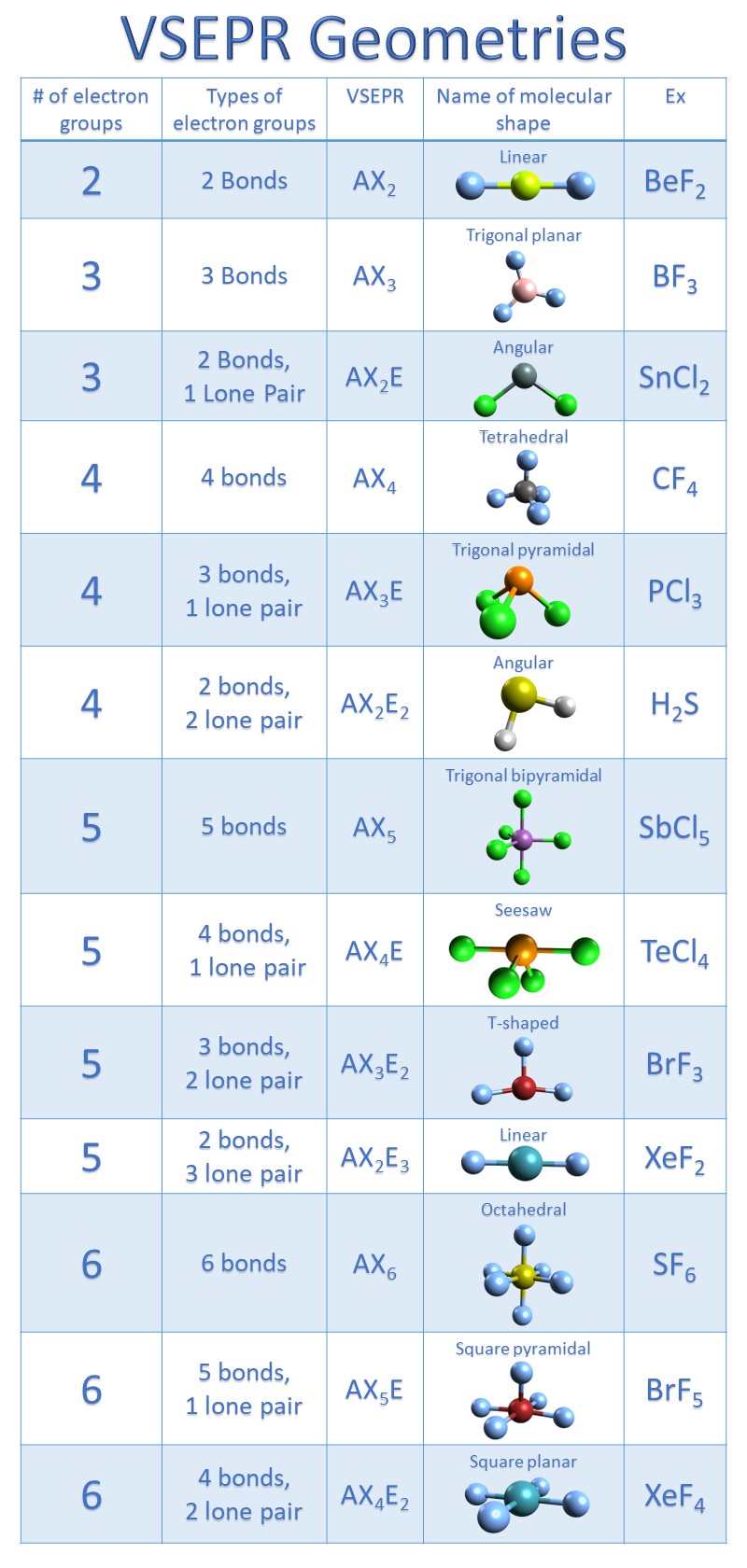
Lewis Structure Of Cocl2
(Valence Electrons Are The Number Of Electrons Present.
(Valence Electrons Are The Electrons That Are Present In.
(Assign Lone Pairs, Radical Electrons, And Atomic Charges Where Appropriate.) Calculate The Electrons Required (Er), Valence Electrons (Ve), And Shared Pairs (Sp).
Draw The Lewis Structure For Cocl2, Including Lone Pairs.
Related Post: