Draw The Lewis Structure For Cs2
Draw The Lewis Structure For Cs2 - Draw a lewis structure for cs2 draw the molecule by placing atoms on the grid and connecting them with bonds. There are three single bonds between the carbon atom (c) and the sulfur atoms (s). Web this is how we draw a complete lewis structure of carbon disulfide. Calculate the total number of valence electrons in cs2: Include all lone pairs of electrons. #1 first draw a rough sketch. Both sulfur atoms have a full octet, and carbon has a formal charge of zero. In order to draw the lewis structure of cs2, first of all you have to find the total number of valence electrons present in the cs2 molecule. Draw the lewis electron dot structures for these molecules,. Web 6 steps to draw the lewis structure of cs2 step #1: Each atom has a full octet, and all valence electrons are involved in bonding. Web the correctly drawn lewis structure for cs2 has no nonbonding electrons. After determining how many valence electrons there are in cs2, place them around the central atom to complete the octets. Web 6 steps to draw the lewis structure of cs2 step #1: Find more. For the cs2 structure use the periodic table to find the total number of valence electrons for. Web the lewis structure for the cs2 molecule can be drawn as follows: Web this widget gets the lewis structure of chemical compounds. Web steps of drawing cs2 lewis structure. Do you know that atoms use hybrid orbitals and not atomic orbitals to. Find more chemistry widgets in wolfram|alpha. 248 views 1 year ago chapter 4 | solution manual for chemistry: Then, the electrons are placed around the atoms to satisfy the octet rule, which states that each atom should have eight electrons in its valence shell. Draw a lewis structure for cs2. Calculate the total number of valence electrons in cs2: Web steps of drawing cs2 lewis structure. Then, the electrons are placed around the atoms to satisfy the octet rule, which states that each atom should have eight electrons in its valence shell. Identify the central atom, which is the least electronegative atom. For the cs2 lewis structure, calculate the total number of valence electrons for the cs2 molecule. Lewis. Do you know that atoms use hybrid orbitals and not atomic orbitals to form chemical bonds in a. Calculate the number of valence electrons: Determine the central metal atom: Distribution of remaining valence electrons: Web drawing the lewis structure for cs 2. Distribution of remaining valence electrons: Each atom has a full octet, and all valence electrons are involved in bonding. We draw lewis structures to predict: Figure out how many electrons the molecule must have, based on the number of valence electrons in each atom. Write the correct skeletal structure for the molecule. 248 views 1 year ago chapter 4 | solution manual for chemistry: Web in short, these are the steps you need to follow for drawing a lewis structure: When drawing the structure of an ion, be sure to add/subtract electrons to account for. We draw lewis structures to predict: Web lewis structure for cs2 (carbon disulfide) commonly tested lewis structures. Total number of valence electrons: Calculate the total number of valence electrons in cs2: Draw a lewis structure for cs2 draw the molecule by placing atoms on the grid and connecting them with bonds. Web the following procedure will give you the correct lewis structure for any molecule or polyatomic ion that has one central atom. Get the free lewis. Web use these steps to correctly draw the cs 2 lewis structure: Draw a lewis structure for cs2. Web to draw the cs2 lewis structure, one must first determine the number of valence electrons for each atom, which is 4 for carbon and 6 for sulfur. * hydrogen atoms are always terminal (only one bond) * put more electronegative elements. There are no nonbonding electrons in the cs2 molecule. The lewis structure for cs2 has no nonbonding electrons. Connect the atoms with dots: Get the free lewis structure finder widget for your website, blog, wordpress, blogger, or igoogle. A video explanation of how to draw the lewis dot structure for carbon disulfide,. Both sulfur atoms have a full octet, and carbon has a formal charge of zero. When drawing the structure of an ion, be sure to add/subtract electrons to account for. Web the lewis structure for the cs2 molecule can be drawn as follows: Web this is how we draw a complete lewis structure of carbon disulfide. Write the correct skeletal structure for the molecule. Web the following procedure will give you the correct lewis structure for any molecule or polyatomic ion that has one central atom. After determining how many valence electrons there are in cs2, place them around the central atom to complete the octets. A video explanation of how to draw the lewis dot structure for carbon disulfide,. Find more chemistry widgets in wolfram|alpha. #1 first draw a rough sketch. Web a step by step guide to draw carbon disulfide, cs2 lewis structure. #2 mark lone pairs on the atoms. * hydrogen atoms are always terminal (only one bond) * put more electronegative elements in terminal positions. Here, the given molecule is cs2 (carbon disulfide). 8 + (2 × × 7) = 22 xef 6: Total number of valence electrons: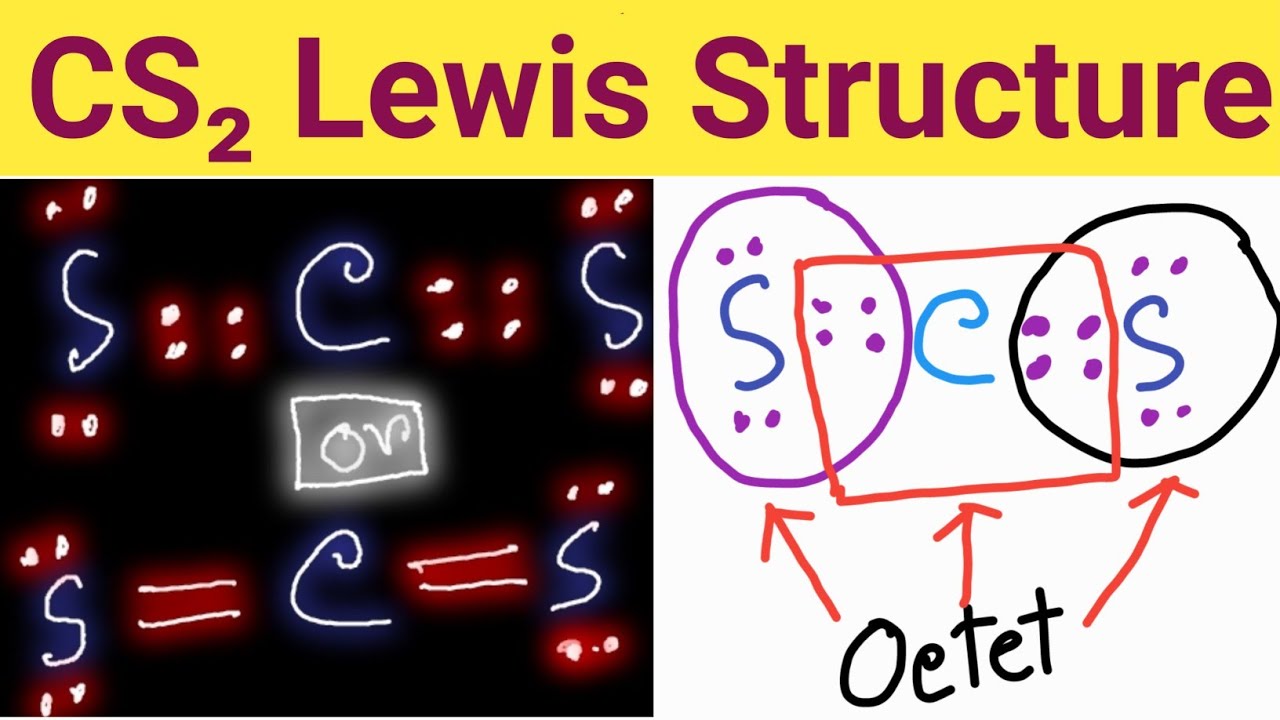
CS2 Lewis Structure Lewis Dot Structure for CS2 Carbon Disulfide
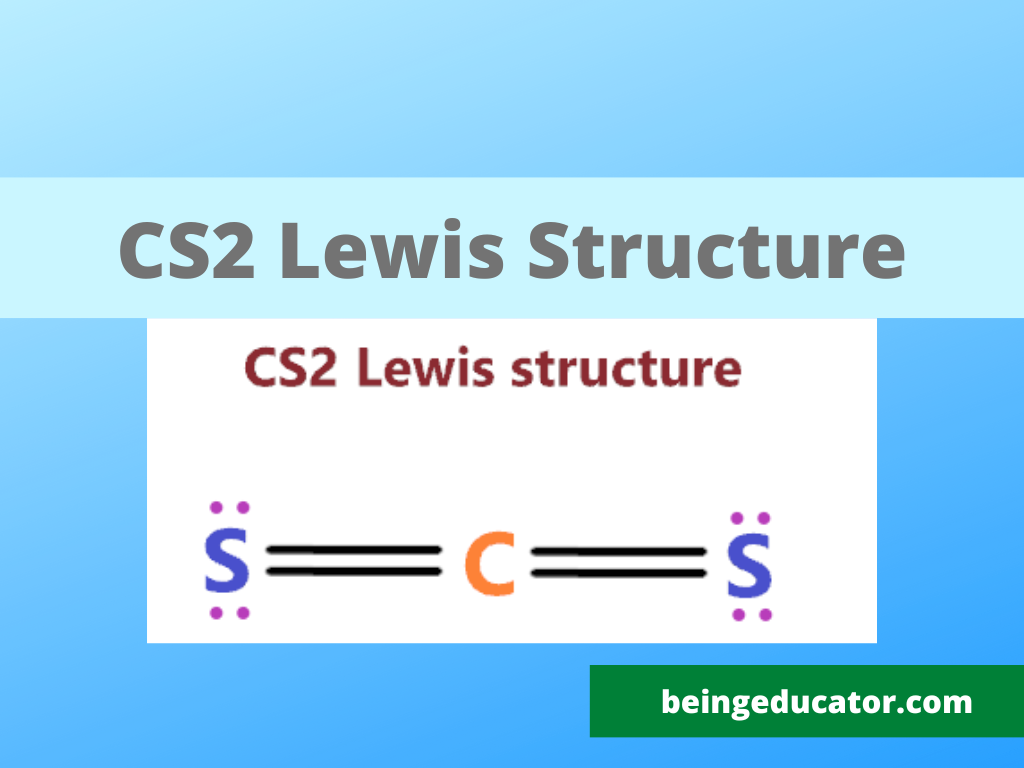
CS2 Lewis Structure Molecular Geometry Polarity Hybridization
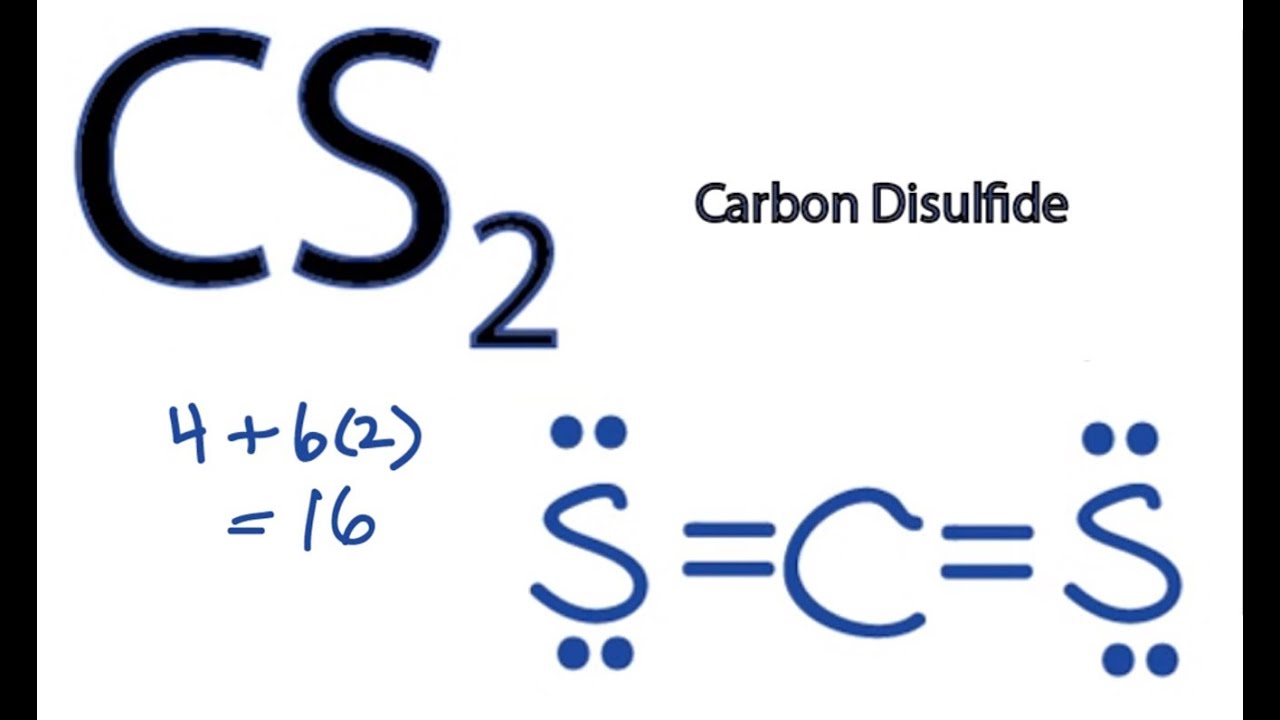
CS2 Lewis Structure How to Draw the Lewis Structure for CS2 YouTube

CS2 Lewis Structure in 4 simple Steps with images What's Insight

CS2 Lewis Structure ,Valence Electrons ,Formal Charge,Polar or Nonpolar

CS2 Lewis Structure ,Valence Electrons ,Formal Charge,Polar or Nonpolar

CS2 Lewis Structure ,Valence Electrons ,Formal Charge,Polar or Nonpolar

How do you draw the Lewis structure of CS2 (Carbon disulfide) YouTube
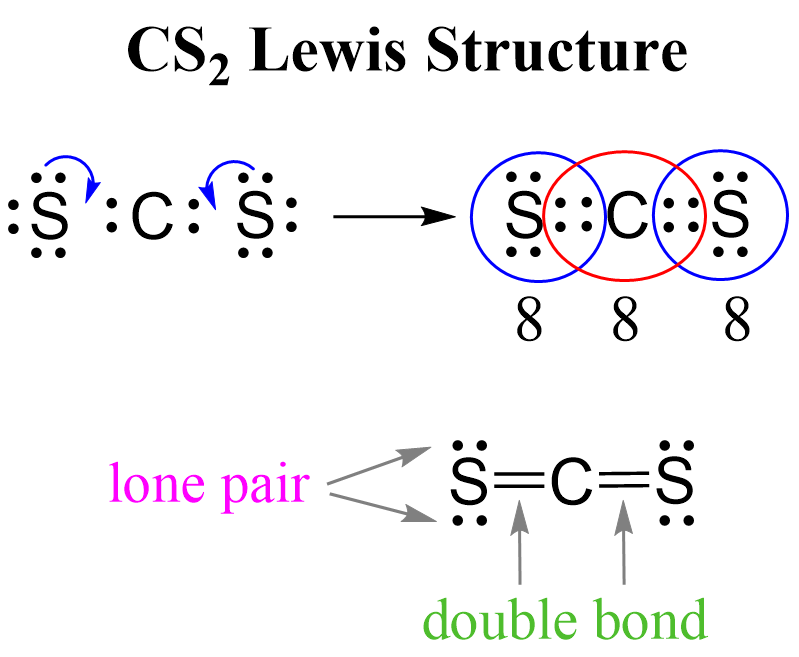
CS2 Lewis Structure, Geometry, and Hybridization Chemistry Steps

Lewis Dot Structure for CS2 Carbon disulfide YouTube
So, Option A Is Incorrect.
In This Case, We Can Condense The Last Few Steps, Since Not All Of Them Apply.
In Order To Find The Total Valence Electrons In Cs2 (Carbon Disulfide) Molecule, First Of All You Should Know The Valence Electrons Present In Carbon Atom As Well As Sulfur Atom.
Web This Widget Gets The Lewis Structure Of Chemical Compounds.
Related Post: