Draw The Lewis Structure Of Xef4 Include Lone Pairs
Draw The Lewis Structure Of Xef4 Include Lone Pairs - Xef4 lewis structure formal charges. Find the total valence electrons in xef4 molecule. Web april 23, 2022 by madhusudan dn. The xeof4 lewis structure refers to the arrangement of atoms and electrons in the xeof4 molecule. Now that we know the valence electrons of xenon tetrafluoride, it will be. Draw the molecule by placing atoms on the canvas and connecting them with bonds. This is a rare example of a noble gas forming a chemical compound. Steps of drawing xef4 lewis structure. When we write the lewis structures for these. Xenon can be the central atom of a molecule by expanding beyond an octet of electrons. Xeof4 is a compound composed of. In order to find the total valence electrons in xef4. Hence there are a total of 36 valence electrons in xef4. Steps of drawing xef4 lewis structure. This is a rare example of a noble gas forming a chemical compound. #1 draw a rough skeleton structure. #2 mention lone pairs on the atoms. Web for the xef4 lewis structure we first count the valence electrons for the xef4 molecule using the periodic table. Xenon (xe) has two lone pairs, and each. Here’s how you can easily draw the xef 4 lewis structure step by step: Web in some hypervalent molecules, such as if 5 and xef 4, some of the electrons in the outer shell of the central atom are lone pairs: Web the lewis structure of xef 2 shows two bonding pairs and three lone pairs of electrons around the xe atom: The xeof4 lewis structure refers to the arrangement of atoms and electrons. Steps of drawing xef4 lewis structure. Xeof4 is a compound composed of. Hence there are a total of 36 valence electrons in xef4. Steps of drawing lewis structure of xef 4. Xef4 lewis structure formal charges. Web for the xef4 lewis structure we first count the valence electrons for the xef4 molecule using the periodic table. Draw lewis structures for the given xenon. Now that we know the valence electrons of xenon tetrafluoride, it will be. Xenon (xe) has two lone pairs, and each. Draw the molecule by placing atoms on the canvas and connecting them. Web the lewis structure of xef4 looks like this: We place three lone pairs of electrons around each f atom, accounting for 12 electrons and giving each f atom 8 electrons. Hence there are a total of 36 valence electrons in xef4. Xenon (xe) sits in the middle, and it's connected to four fluorine atoms (f) with single bonds. #2. When we write the lewis structures for these. #1 draw a rough skeleton structure. Xenon (xe) sits in the middle, and it's connected to four fluorine atoms (f) with single bonds. Web the lewis structure of xef 2 shows two bonding pairs and three lone pairs of electrons around the xe atom: Draw lewis structures for the given xenon. Xeof4 is a compound composed of. Steps of drawing lewis structure of xef 4. Xenon (xe) has two lone pairs, and each. Find the total valence electrons in xef4 molecule. Draw the molecule by placing atoms on the canvas and connecting them with bonds. Now that we know the valence electrons of xenon tetrafluoride, it will be. Web the lewis structure of xef4 looks like this: Web write the lewis structure for xef4. #1 draw a rough skeleton structure. Web in some hypervalent molecules, such as if 5 and xef 4, some of the electrons in the outer shell of the central atom are. Xenon (xe) has two lone pairs, and each. Web the lewis structure of xef 2 shows two bonding pairs and three lone pairs of electrons around the xe atom: Web the lewis structure indicates that each cl atom has three pairs of electrons that are not used in bonding (called lone pairs) and one shared pair of electrons (written between.. Hence there are a total of 36 valence electrons in xef4. #2 mention lone pairs on the atoms. Now that we know the valence electrons of xenon tetrafluoride, it will be. Once we know how many valence electrons there are in xef4. Web april 23, 2022 by madhusudan dn. Web in the xef4 lewis structure, there are more than 8 valence electrons which include 2 lone pairs and the bonding electrons. The xeof4 lewis structure refers to the arrangement of atoms and electrons in the xeof4 molecule. Xeof4 is a compound composed of. We place three lone pairs of electrons around each f atom, accounting for 12 electrons and giving each f atom 8 electrons. Steps of drawing xef4 lewis structure. Include all lone pairs of electrons. Web the lewis structure indicates that each cl atom has three pairs of electrons that are not used in bonding (called lone pairs) and one shared pair of electrons (written between. #1 draw a rough skeleton structure. Draw the molecule by placing atoms on the canvas and connecting them with bonds. Xenon (xe) sits in the middle, and it's connected to four fluorine atoms (f) with single bonds. Web write the lewis structure for xef4.
XeF4 Lewis Structure Drawing easy steps,Hybridization,shape
![Molecular Geometry of XeF4 [with video and free study guide]](https://aceorganicchem.com/chemistry/wp-content/uploads/2023/05/XeF4-lewis-dot-2.jpg)
Molecular Geometry of XeF4 [with video and free study guide]

XeF4 Lewis structure, Molecular geometry, Bond angle, Shape

XeF4 Lewis Structure Drawing easy steps,Hybridization,shape

Draw The Lewis Structure For The Following Molecule Xef4

XeF4 Lewis Structure How to Draw the Lewis Structure for XeF4 YouTube

XeF4 Lewis Structure Drawing easy steps,Hybridization,shape
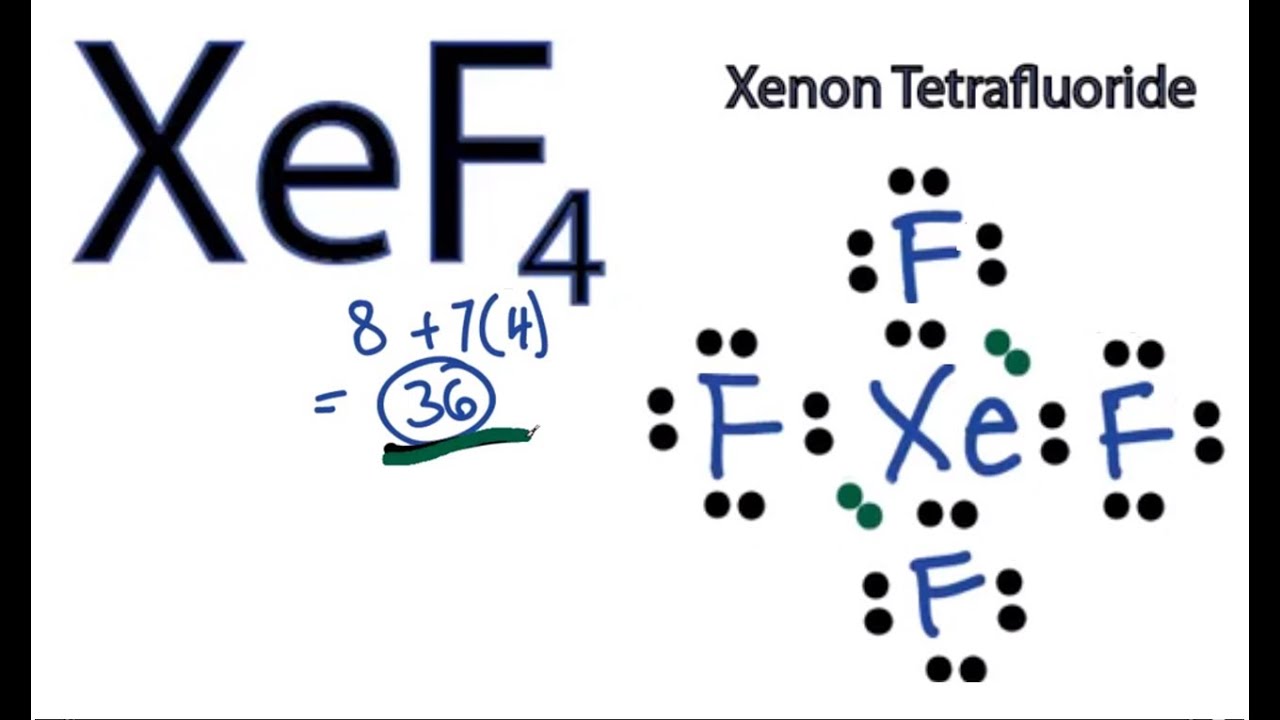
XeF4 Lewis Structure How to Draw the Lewis Structure for XeF4 YouTube

XeF4 Lewis Structure Drawing easy steps,Hybridization,shape

Xeof4 Lewis Structure Geometry Hybridization And Polarity
How Do You Draw The Lewis Structure For #Opbr_3#?
Xef4 Lewis Structure Formal Charges.
Web In Some Hypervalent Molecules, Such As If 5 And Xef 4, Some Of The Electrons In The Outer Shell Of The Central Atom Are Lone Pairs:
Xenon (Xe) Has Two Lone Pairs, And Each.
Related Post: