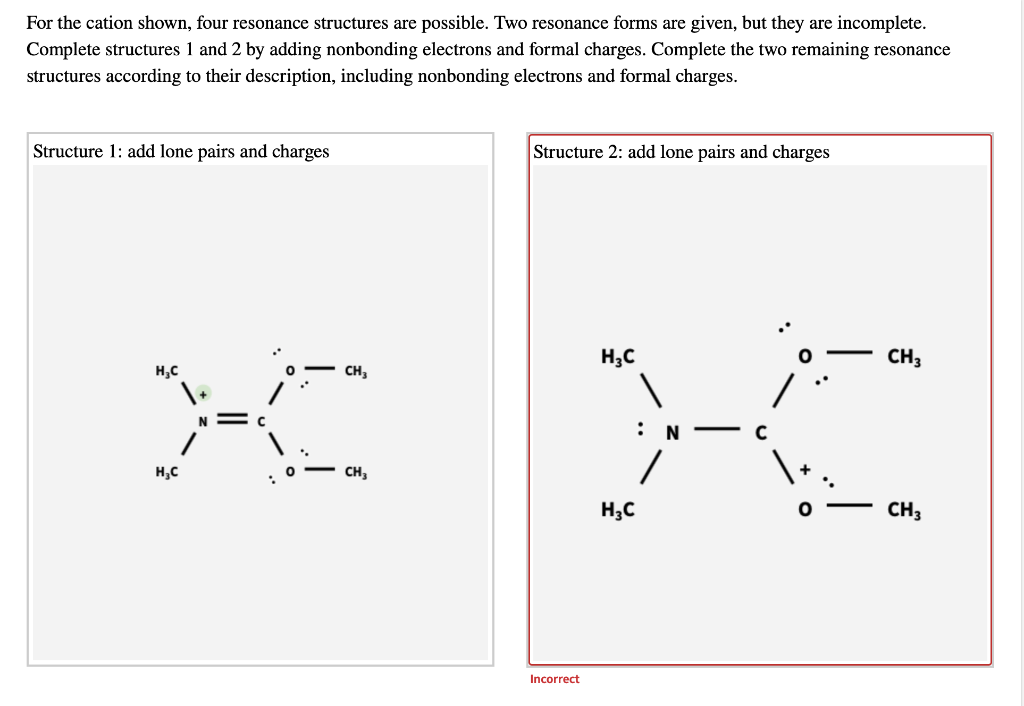
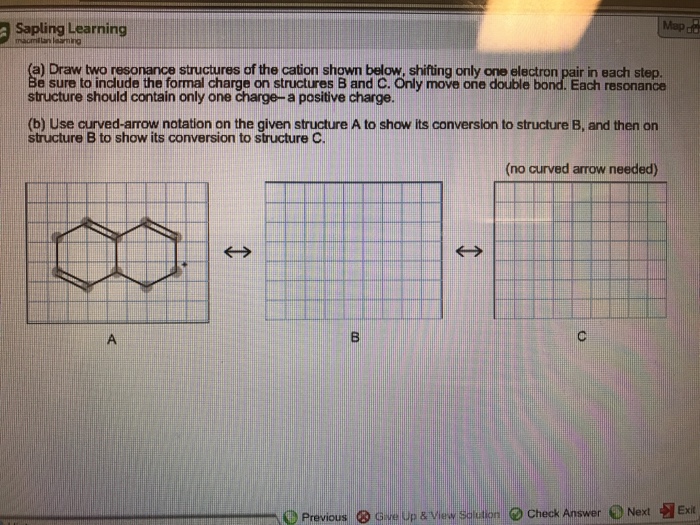
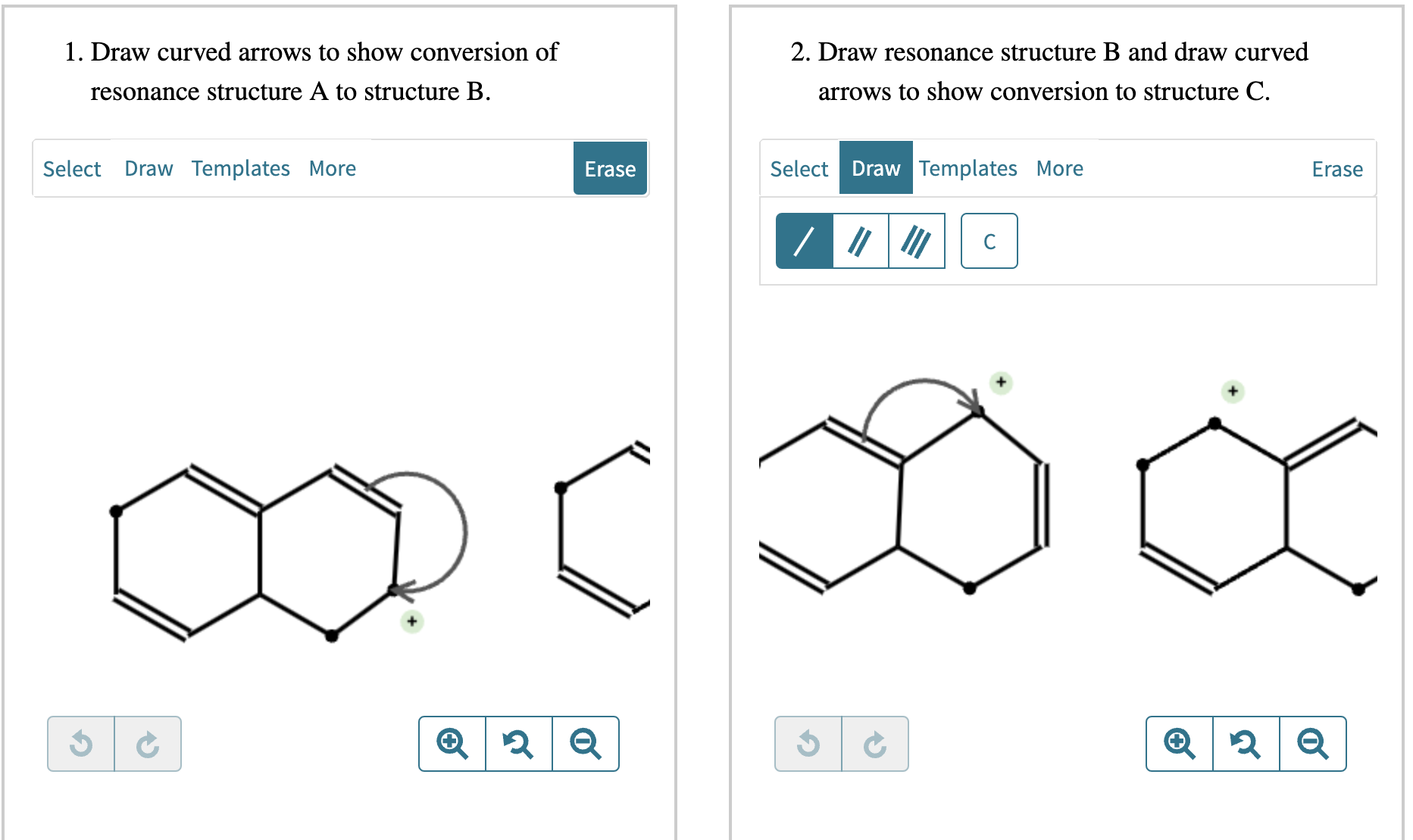
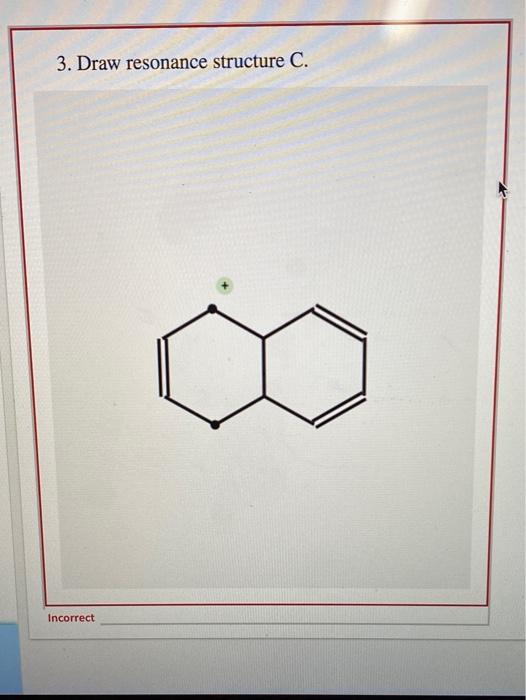
Draw Two Resonance Structures Of The Cation Shown
Draw Two Resonance Structures Of The Cation Shown - Web in the case of the acetate ion and the allyl cation, both resonance forms are equal in energy, so the “hybrid” is a 1:1 mixture of the two. Web how to draw resonance structures. Be sure to include the formal charge on structures b and c. We want to only move one pair of electrons because we want to show our two vesinet structures. Be sure to include the formal charge on structures b and c. Draw two resonance structures of the cation shown, shifting only one electron pair in each step. Web draw the resonance structures of molecules or ions that exhibit delocalization. Web explain the concept of resonance and draw lewis structures representing resonance forms for a given molecule Some molecules have two or more chemically equivalent lewis electron structures, called resonance structures. Web these two drawings are an example of what is referred to in organic chemistry as resonance contributors: Two or more different lewis structures depicting the same. Be sure to include the formal charge on structures b and c. Web draw the resonance structures of molecules or ions that exhibit delocalization. Sometimes one dot structures is not enough to completely describe a molecule or an ion, sometimes you need two or more, and here's an example: Introducing curved. Web (a) draw two resonance structures of the cation shown below, shifting only one electron pair in each step. Determine the relative stability of resonance structures using a set of rules. Web the thiocyanate ion (scn −), which is used in printing and as a corrosion inhibitor against acidic gases, has at least two possible lewis electron structures. We want. Web draw the resonance structures of molecules or ions that exhibit delocalization. Calculate the total number of valence electrons from each atom. Web (a) draw two resonance structures of the cation shown below, shifting only one electron pair in each step. Sometimes one dot structures is not enough to completely describe a molecule or an ion, sometimes you need two. Web these two drawings are an example of what is referred to in organic chemistry as resonance contributors: Web (a) draw two resonance structures of the cation shown below, shifting only one electron pair in each step. Web resonance contributors do not have to be equivalent. Web how to draw resonance structures. Remember that when a carbon has a positive. Web in the case of the acetate ion and the allyl cation, both resonance forms are equal in energy, so the “hybrid” is a 1:1 mixture of the two. Web (a) draw two resonance structures of the cation shown below, shifting only one electron pair in each step. Web how to draw resonance structures. Be sure to include the formal. Web resonance contributors do not have to be equivalent. Web how to draw resonance structures. We want to only move one pair of electrons because we want to show our two vesinet structures. Web in chemistry, resonance, also called mesomerism, is a way of describing bonding in certain molecules or polyatomic ions by the combination of several contributing structures into.. Web (a) draw two resonance structures of the cation shown below, shifting only one electron pair in each step. We can convert one resonance form into. Web explain the concept of resonance and draw lewis structures representing resonance forms for a given molecule Web these two drawings are an example of what is referred to in organic chemistry as resonance. Web draw the resonance structures of molecules or ions that exhibit delocalization. Web the thiocyanate ion (scn −), which is used in printing and as a corrosion inhibitor against acidic gases, has at least two possible lewis electron structures. Sometimes one dot structures is not enough to completely describe a molecule or an ion, sometimes you need two or more,. Be sure to include the formal charge on structures b and c. Web (a) draw two resonance structures of the cation shown below, shifting only one electron pair in each step. Introducing curved arrows, a tool for showing the movement of electrons between resonance structures. Web in the case of the acetate ion and the allyl cation, both resonance forms. Determine the relative stability of resonance structures using a set of rules. Web for each example, specify whether the two structures are resonance contributors to the same resonance hybrid. (a) draw two resonance structures of the cation shown, shifting only one electron pair in each step. However, this is only rarely the case. Web in chemistry, resonance, also called mesomerism,. Calculate the total number of valence electrons from each atom. Remember that when a carbon has a positive charge, it has 3. Web (a) draw two resonance structures of the cation shown below, shifting only one electron pair in each step. However, this is only rarely the case. (a) draw two resonance structures of the cation shown, shifting only one electron pair in each step. Determine the relative stability of resonance structures using a set of rules. Web (a) draw two resonance structures of the cation shown below, shifting only one electron pair in each step. Web resonance contributors do not have to be equivalent. Web these two drawings are an example of what is referred to in organic chemistry as resonance contributors: Web explain the concept of resonance and draw lewis structures representing resonance forms for a given molecule Introducing curved arrows, a tool for showing the movement of electrons between resonance structures. Be sure to include the formal charge on structures b and c. Web draw the resonance structures of molecules or ions that exhibit delocalization. Web draw the resonance structures of molecules or ions that exhibit delocalization. We can convert one resonance form into. Web in chemistry, resonance, also called mesomerism, is a way of describing bonding in certain molecules or polyatomic ions by the combination of several contributing structures into.draw two resonance structures of the cation shown below
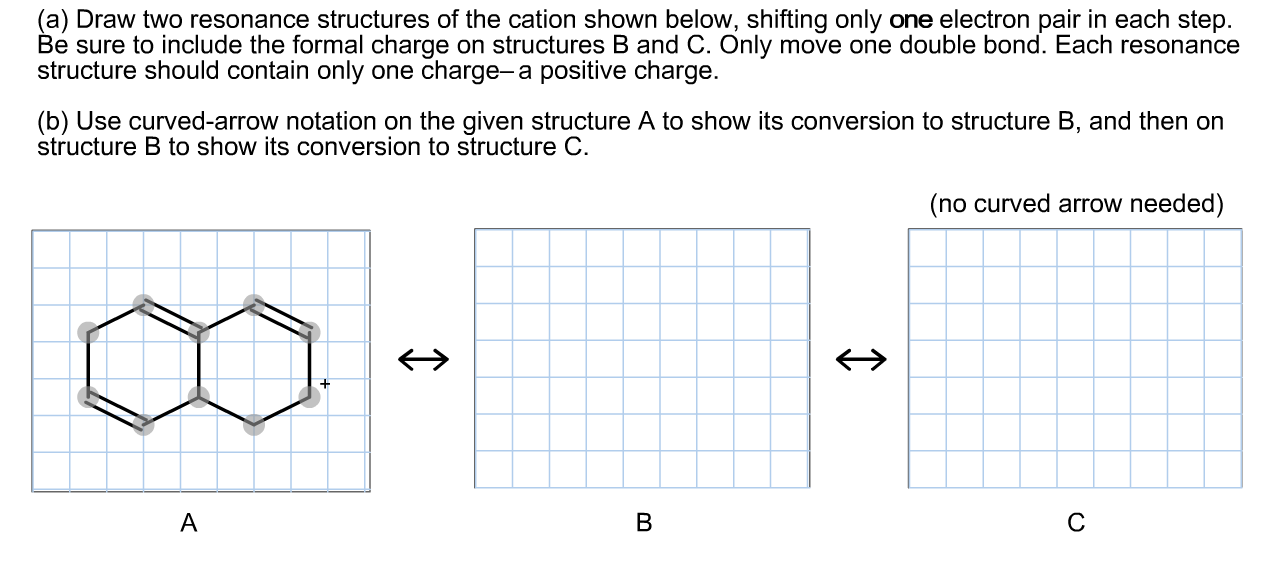
Solved (a) Draw two resonance structures of the cation shown
Solved Draw Two Resonance Structures Of The Cation Shown
Solved Draw two resonance structures of the cation shown,
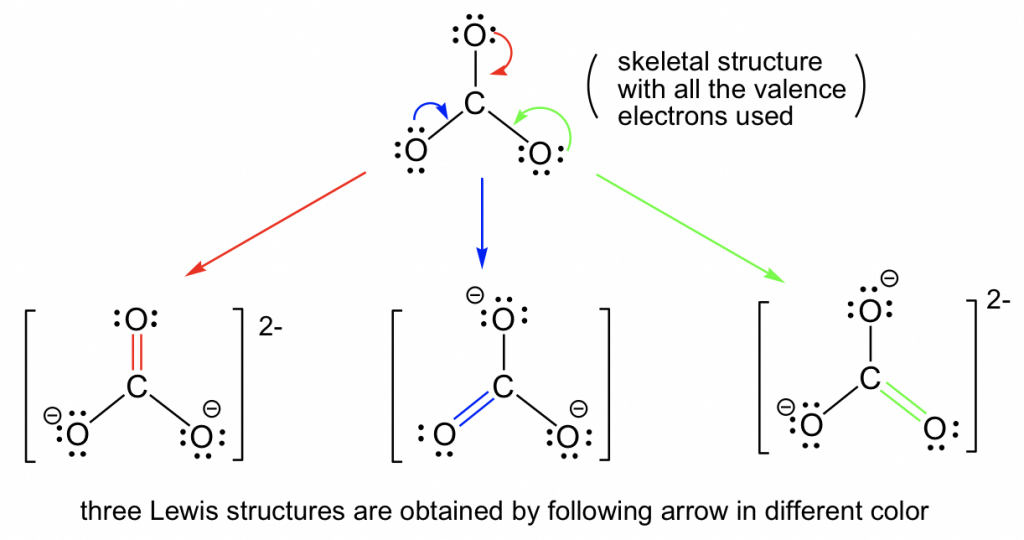
How To Draw Resonance Structures Foreversalary
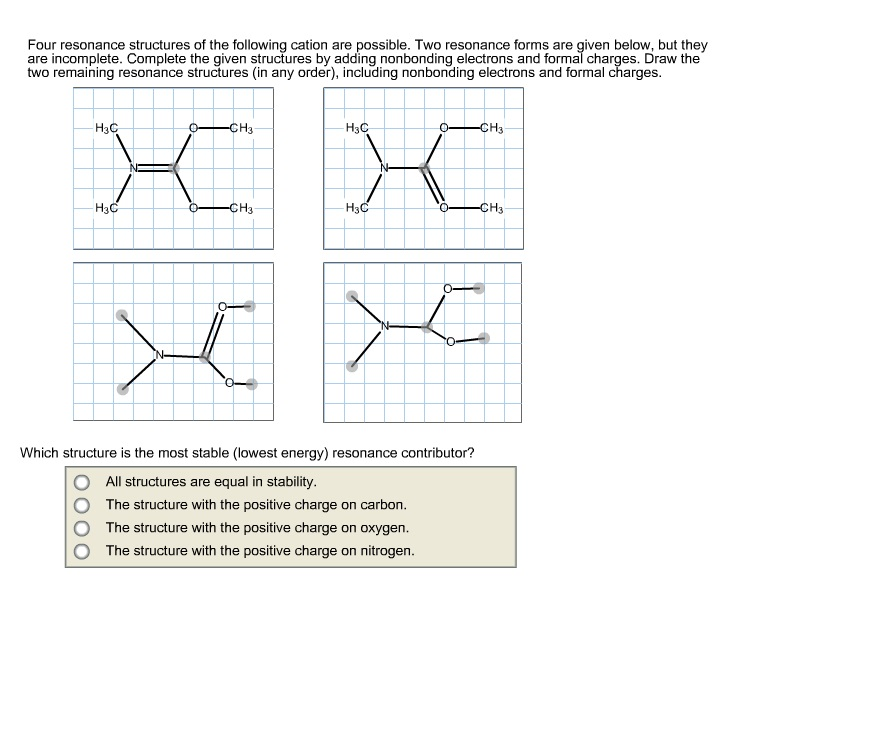
draw two resonance structures of the cation shown below
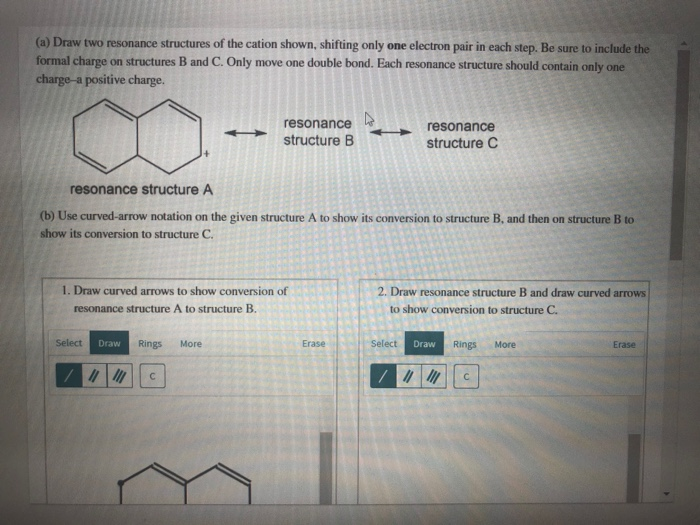
draw two resonance structures of the cation shown below blackbodyart
Solved (a) Draw two resonance structures of the cation

(a) Draw two resonance structures of the cation shown below, shifting
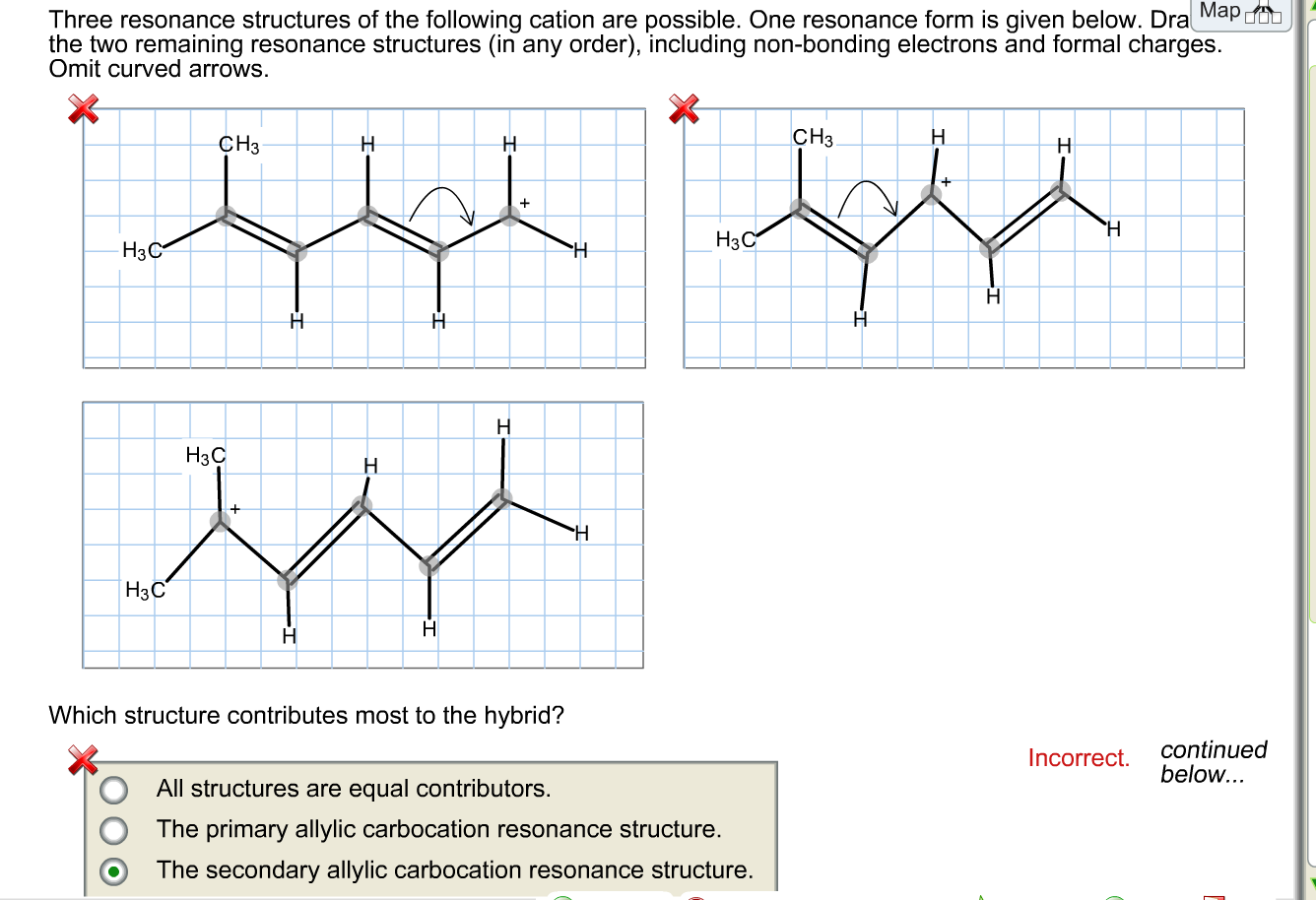
draw two resonance structures of the cation shown below
Determine The Relative Stability Of Resonance Structures Using A Set Of Rules.
Web How To Draw Resonance Structures.
Web The Thiocyanate Ion (Scn −), Which Is Used In Printing And As A Corrosion Inhibitor Against Acidic Gases, Has At Least Two Possible Lewis Electron Structures.
Sometimes One Dot Structures Is Not Enough To Completely Describe A Molecule Or An Ion, Sometimes You Need Two Or More, And Here's An Example:
Related Post: