Drawing Of Covalent Bond
Drawing Of Covalent Bond - Examples for drawing lewis structures for covalent bonds. 1.31 explain the formation of simple molecular, covalent substances, using dot and cross diagrams,. Web how to draw covalent bonding molecules. Web a covalent bond is a chemical bond between two atoms where they share one or more pairs of electrons. 4.7k views 7 years ago 1d: The following procedure can be used to draw lewis structure for simple molecules. Lewis structures are representations of molecules that include not only what atoms are present in the molecule but also how the atoms are connected. Drawing lewis structures for molecules with one central atom: Web covalent bonds between different atoms. Determine the total number of valence electrons in the molecule or ion. Web draw lewis structures for covalent compounds. Exercise \(\pageindex{1}\) rules for drawing covalent lewis structures; The only thing smaller than atoms is their subatomic particles; A chemical bond resulting from two atoms sharing one or more pairs of electrons. 1.31 explain the formation of simple molecular, covalent substances, using dot and cross diagrams,. Illustrate covalent bond formation with lewis electron dot diagrams. Given descriptions, diagrams, scenarios, or chemical symbols, students will model covalent bonds using electron dot formula (lewis structures). A chemical bond resulting from two atoms sharing one or more pairs of electrons. Web we refer to this as a pure covalent bond. Lewis structures are representations of molecules that include not. Given descriptions, diagrams, scenarios, or chemical symbols, students will model covalent bonds using electron dot formula (lewis structures). 1.31 explain the formation of simple molecular, covalent substances, using dot and cross diagrams,. Ionic bonding typically occurs when it is easy for one atom to lose one or more electrons, and for another atom to gain one or more electrons. Exercise. Hydrogen (h 2 ), chlorine (cl 2 ), oxygen (o 2 ), nitrogen (n 2 ), hydrogen chloride (hcl), water (h 2 o), ammonia (nh 3) and methane (ch 4) the correct dot and cross diagrams for these molecules are shown below: Exercise \(\pageindex{1}\) rules for drawing covalent lewis structures; Examples for drawing lewis structures for covalent bonds. Ionic bonding. Each h atom starts with a single electron in its valence shell: 0:08 introduction 0:39 h2 1:25 hcl. Web a covalent bond is formed between two atoms by sharing electrons. The number of bonds an element forms in a covalent compound is determined by the number of electrons it needs to reach octet. Hydrogen (h 2 ), chlorine (cl 2. In any molecule or ion with the general formula abn , the unique atom (a) is in the center and all of the b atoms are attached to a. Drawing lewis dot structures for polyatomic ions. Electrons shared in pure covalent bonds have an equal probability of being near each nucleus. Web how to draw a lewis structure. How can. A lewis diagram shows how the valence electrons are distributed around the atoms in a molecule. Rules for writing covalent formulas; Several socratic answers give the procedure. Covalent bonds usually form between nonmetals. Determine the total number of valence electrons in the molecule or ion. Given descriptions, diagrams, scenarios, or chemical symbols, students will model covalent bonds using electron dot formula (lewis structures). The two h atoms can share their electrons: Web a single line indicates a bond between two atoms (i.e., involving one electron pair), double lines (=) indicate a double bond between two atoms (i.e., involving two electron pairs), and triple lines (≡). Drawing lewis structures for molecules with one central atom: Web a covalent bond is formed between two atoms by sharing electrons. Exercise \(\pageindex{1}\) rules for drawing covalent lewis structures; Web define covalent bond. The number of bonds an element forms in a covalent compound is determined by the number of electrons it needs to reach octet. Practice with drawing lewis structures. Web how to draw a lewis structure. Web draw lewis structures for covalent compounds. Shared pairs of electrons are drawn as lines between atoms, while lone pairs of electrons are drawn as dots next to atoms. Atoms form double or triple covalent bonds if they can attain a noble gas structure by doing so. Covalent bonds usually form between nonmetals. Ionic bonding typically occurs when it is easy for one atom to lose one or more electrons, and for another atom to gain one or more electrons. These structural images are named after gilbert lewis, the american chemist who first proposed that covalent molecules could be represented visually. Drawing lewis dot structures for polyatomic ions. The number of bonds an element forms in a covalent compound is determined by the number of electrons it needs to reach octet. Web how to draw covalent bonding molecules. Rules for writing covalent formulas; The only thing smaller than atoms is their subatomic particles; Shared pairs of electrons are drawn as lines between atoms, while lone pairs of electrons are drawn as dots next to atoms. 4.7k views 7 years ago 1d: H forms only one bond because it needs only two electrons. Usually, sharing electrons gives each atom a full valence shell and makes the resulting compound more stable than its constituent atoms are on their own. Given descriptions, diagrams, scenarios, or chemical symbols, students will model covalent bonds using electron dot formula (lewis structures). In the case of cl 2, each atom starts off with seven valence electrons, and each cl shares one electron with the other, forming one covalent bond: Web a covalent bond is a chemical bond between two atoms where they share one or more pairs of electrons. Draw lewis structures for covalent compounds.
How To Draw Covalent Bonds
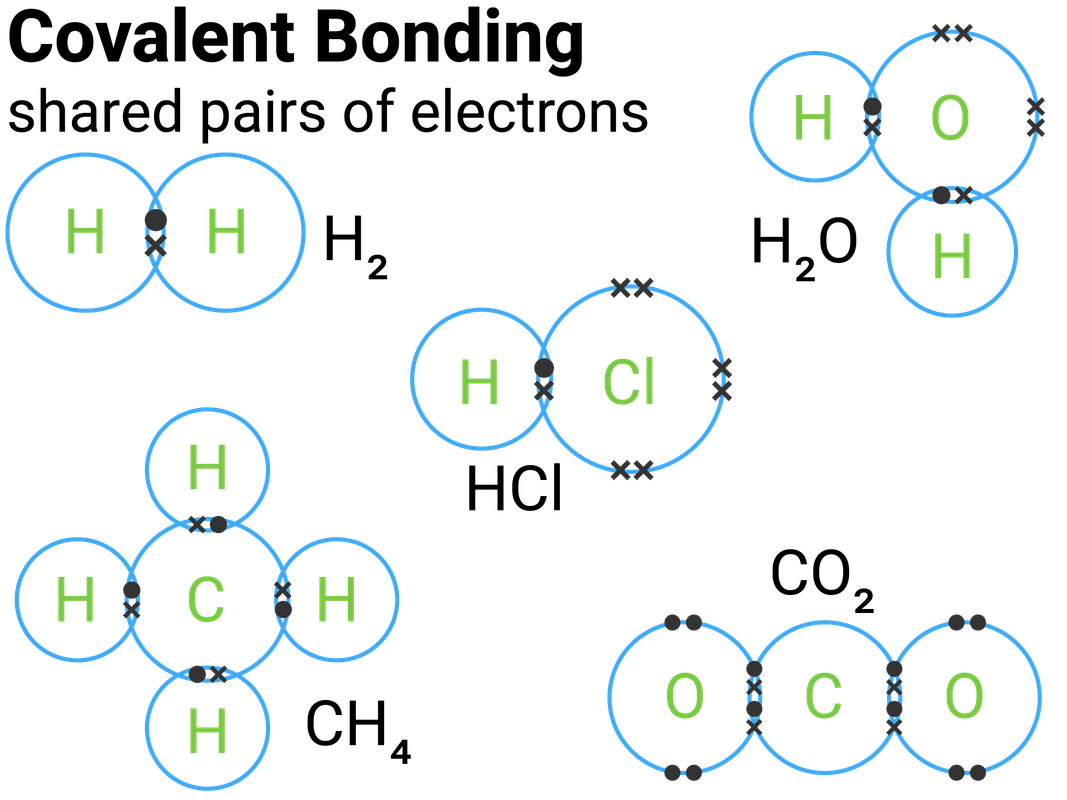
Chemical Bonds, Ionic, Covalent and Metallic AQA C2 revisechemistry.uk

How is a covalent bond formed

Covalent Bonding The Science and Maths Zone

Covalent Bonding The Science and Maths Zone

Covalent bonding Dot and cross diagrams Graphite
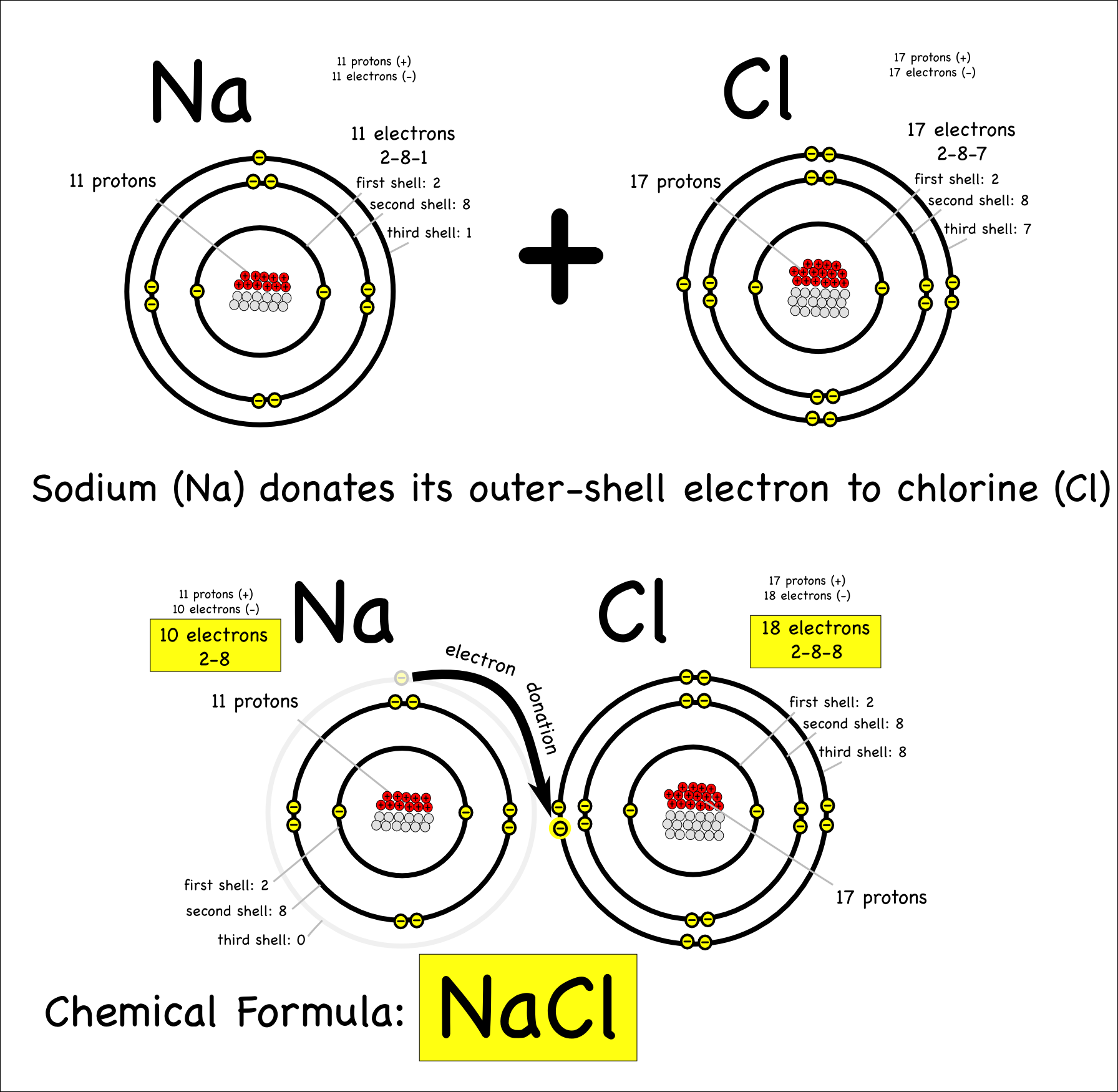
Introducing Covalent Bonding Montessori Muddle
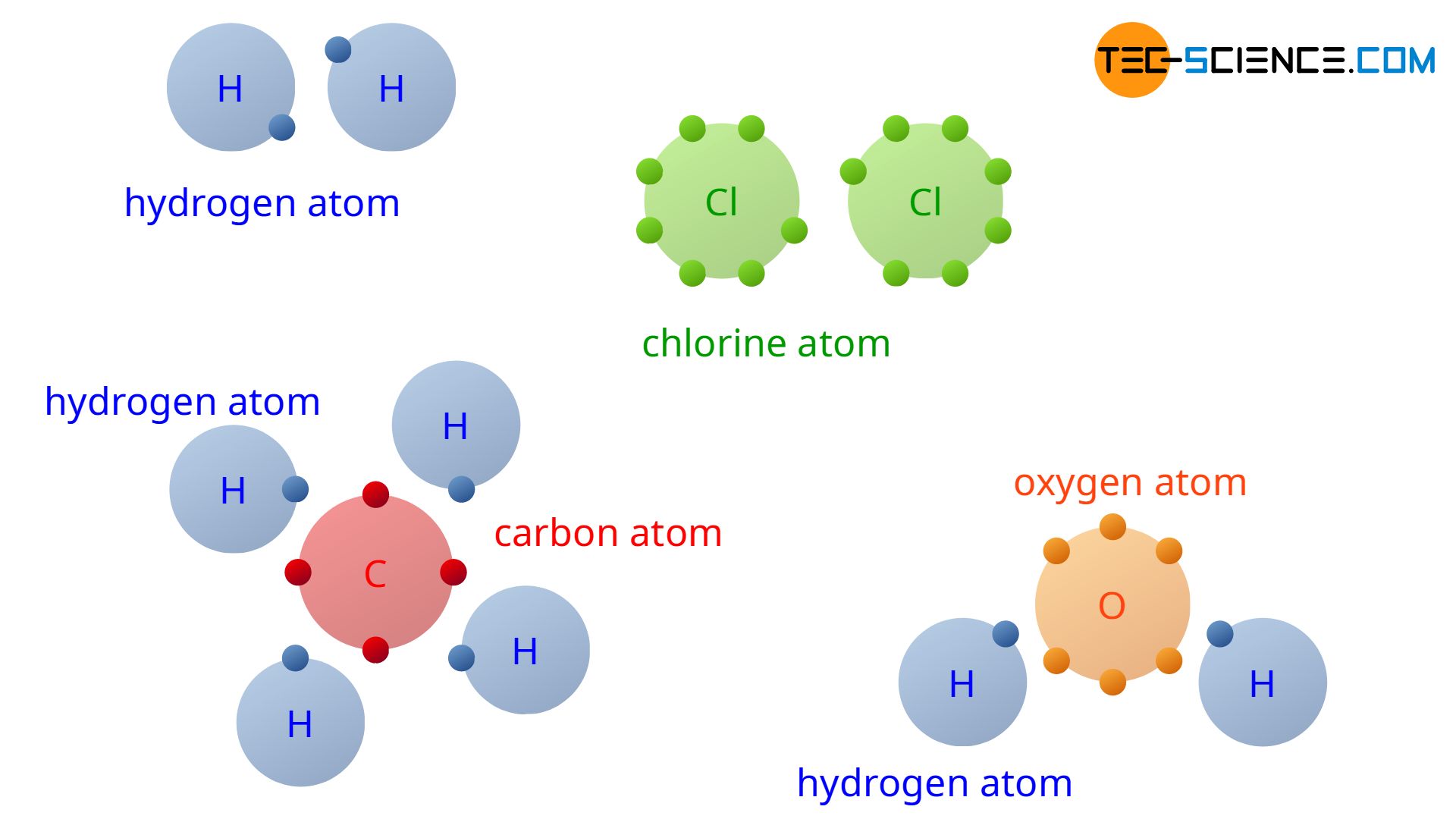
Covalent bonding tecscience
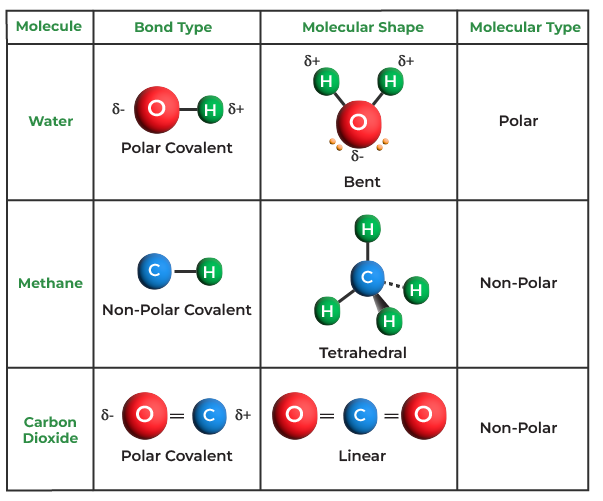
Covalent Bond Definition, Examples, Types, Properties, & FAQs
Covalent bond (covalency) and its type Overall Science
Practice With Drawing Lewis Structures.
Exercise \(\Pageindex{1}\) Rules For Drawing Covalent Lewis Structures;
Web A Single Line Indicates A Bond Between Two Atoms (I.e., Involving One Electron Pair), Double Lines (=) Indicate A Double Bond Between Two Atoms (I.e., Involving Two Electron Pairs), And Triple Lines (≡) Represent A Triple Bond, As Found, For Example, In.
0:08 Introduction 0:39 H2 1:25 Hcl.
Related Post: