How To Draw Mo Diagrams
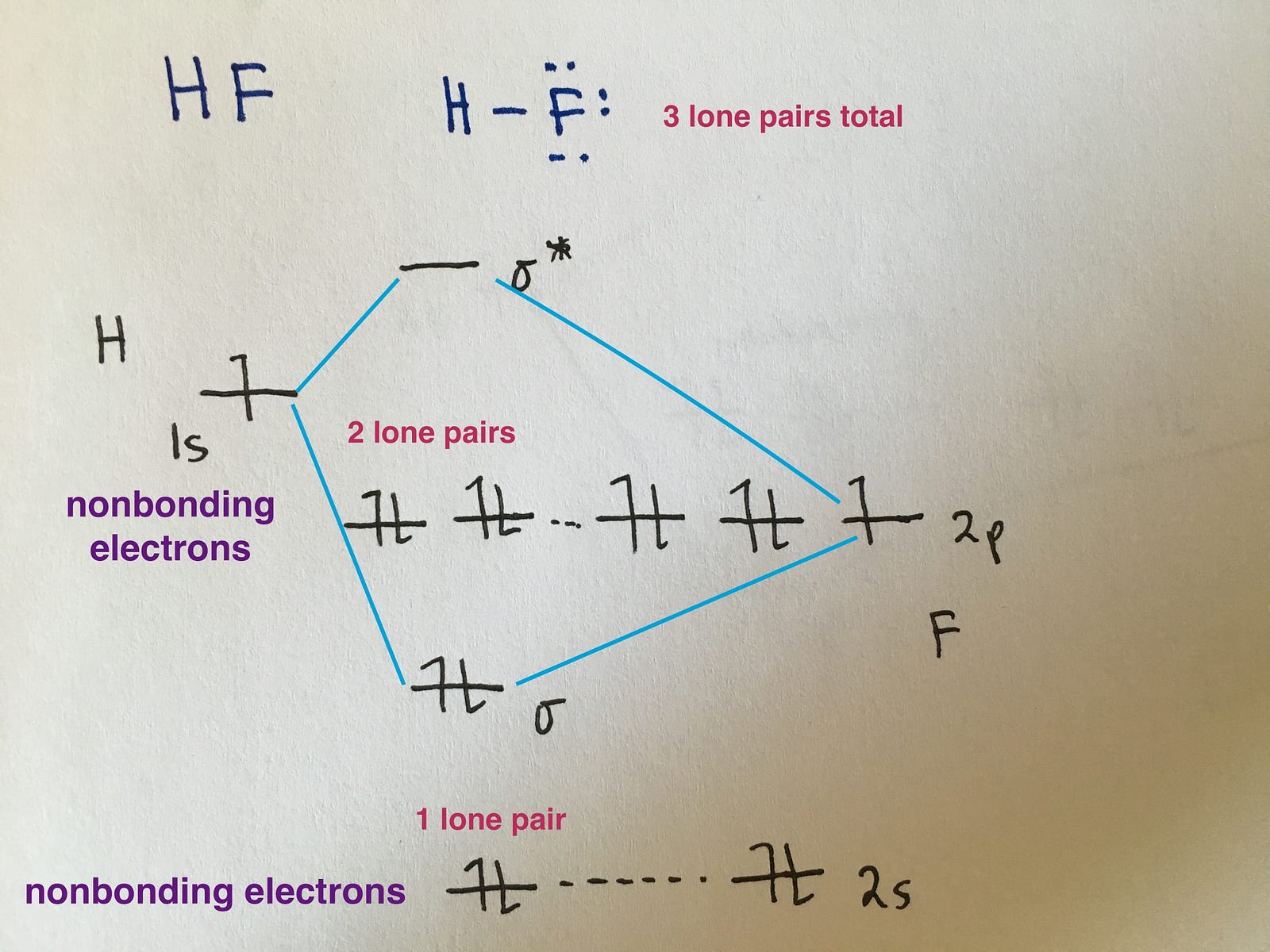
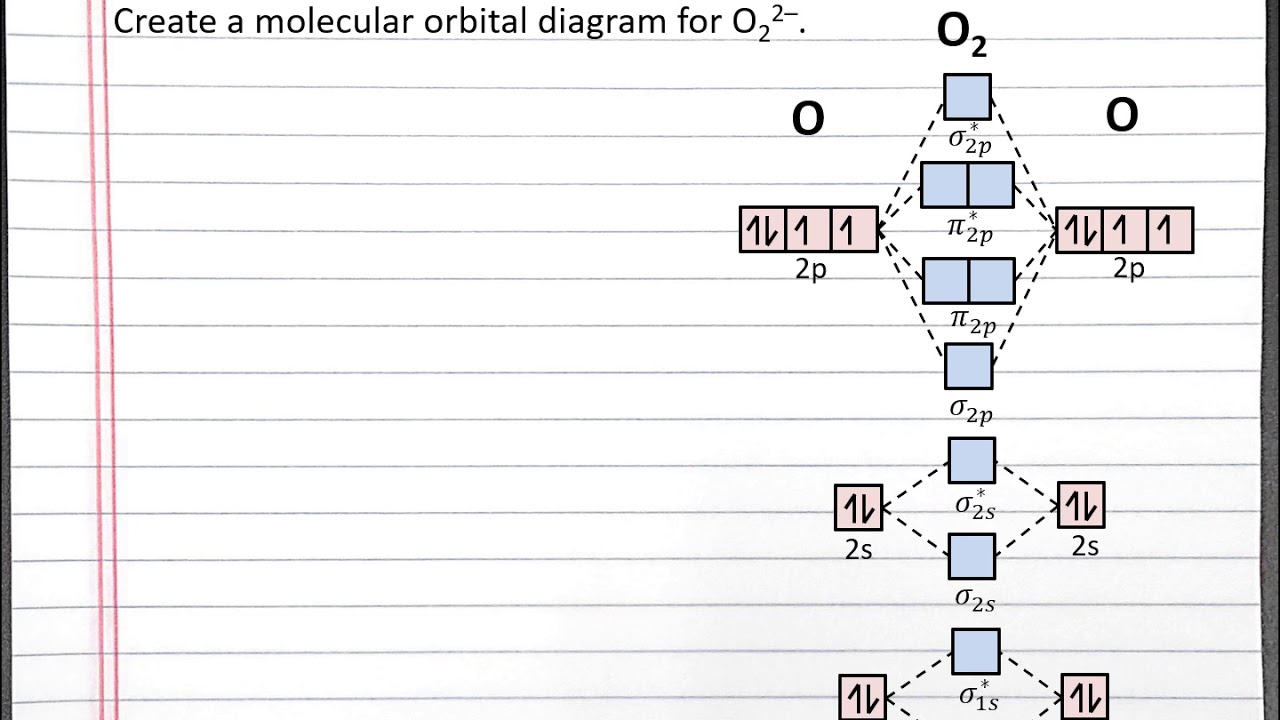
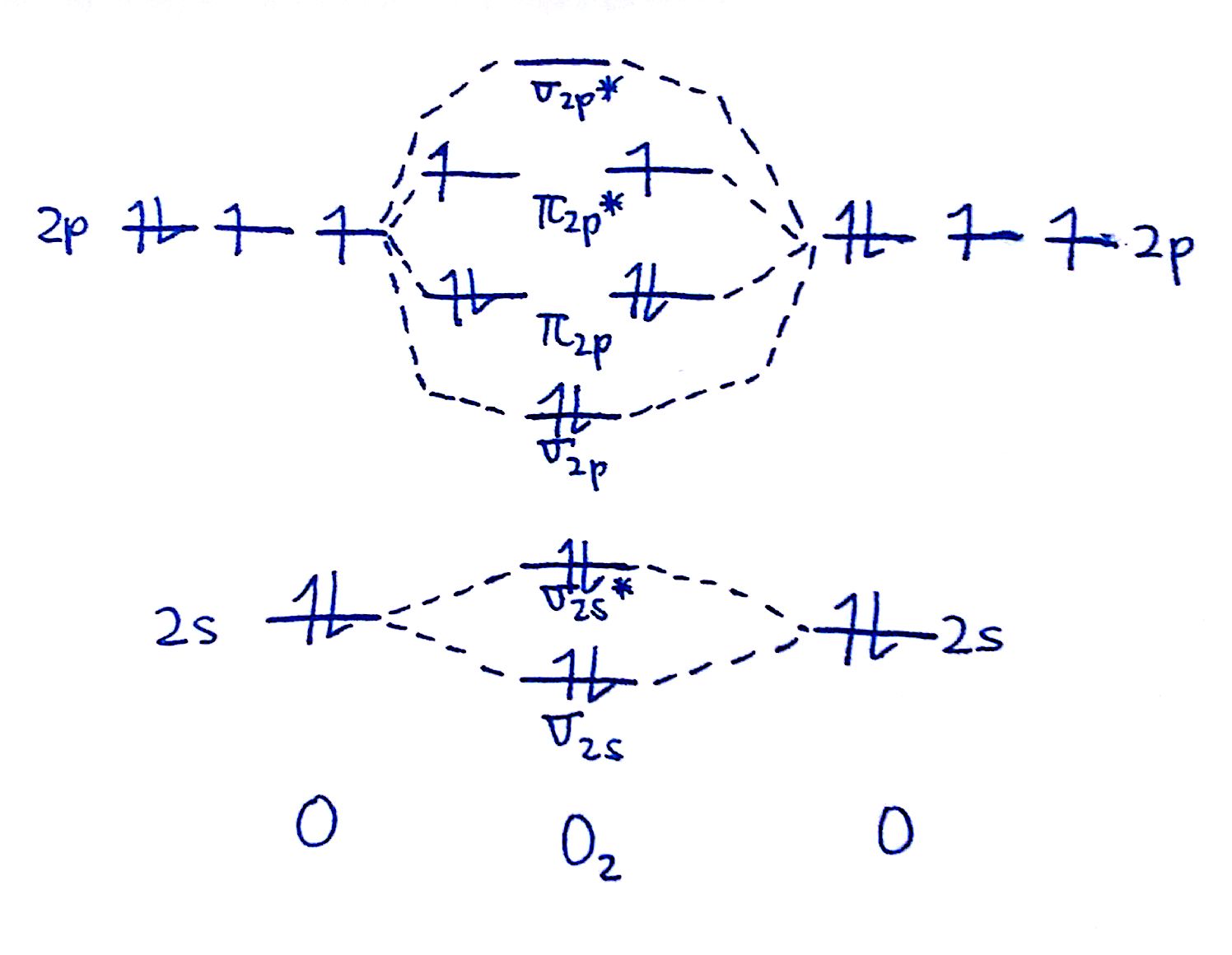
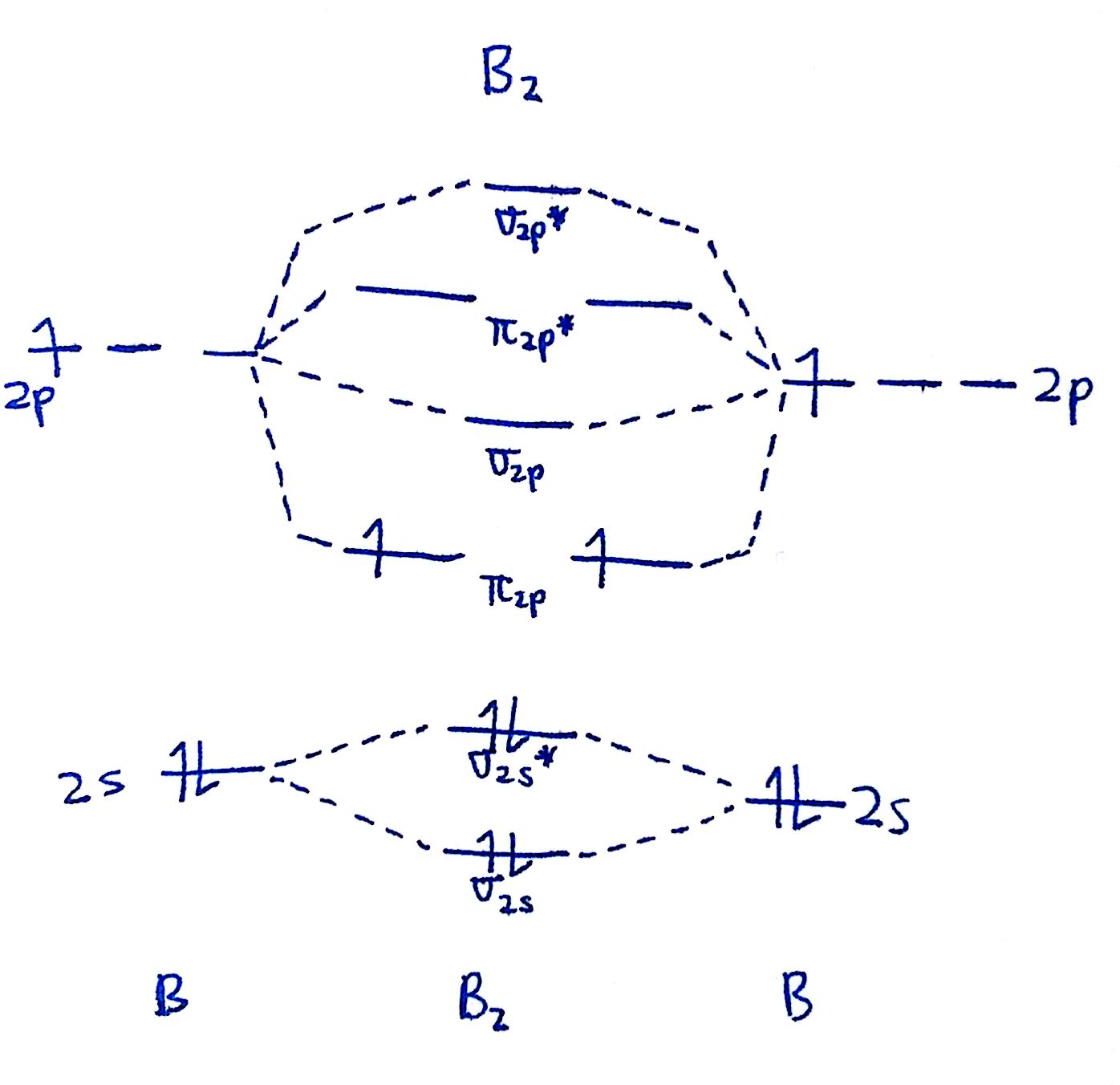
How To Draw Mo Diagrams - As a rule of thumb, a bond order = 1 equates to a single bond, a bond order = 2 equates to a double bond, etc. That is, it's `o_2` with `1` missing electron. Now let's put these ideas together to make an mo diagram for hf. It also illustrates the operations and attributes of the classes. Web molecular orbital diagrams. H has a 1s orbital. `o_2^+` is just the ionized form of `o_2`; Molecular orbital diagrams are complex, involving two additional orbitals, electronegativity, atomic symmetries and atomic energies. In the event of a discrepancy, the official drawing results shall prevail. Molecular orbitals for a simple pi bond; Draw the mo for o 2: They are usually used to explore domain concepts, understand software requirements and. Web similar to atomic orbitals, we can write electron configuration energy diagrams for molecular orbitals (figure 9.20 “ hydrogen molecular orbital electron configuration energy diagram”).notice that the atomic orbitals of each atom are written on either side, and the newly formed molecular. Molecular orbitals for a simple pi bond; We are only going to consider valence orbitals. Web for more complicated molecules, it is better to use the procedure given earlier: The mo diagram will be the same as the mo diagram of `o_2`, except with `1` less electron. Web molecular orbital diagrams. Web for more complicated molecules, it is better to use the procedure given earlier: Official winning numbers are those selected in the respective drawings and recorded under the observation of an independent accounting firm. H has a 1s orbital. Determine the total number of valence electrons in the he 2 2 + ion. Molecular orbitals for a simple pi bond; The symmetry of group orbitals is determined by reducing a reducible representation of the orbitals in question. Two for the allyl cation, three for the allyl radical, and four for the allyl anion. Valence bond theory and molecular. Now let's put these ideas together to make an mo diagram for hf. Web mo diagram for hf. Molecular orbitals for a simple pi bond; Draw the mo diagram for `o_2^+` this is a bit of a curveball, but a perfectly valid problem. It describes the formation of bonding and antibonding molecular o. Try the following mo's on your own, and then check with the answers provided. Valence bond theory and molecular. They are usually used to explore domain concepts, understand software requirements and. Recall that a cation indicates a loss of `1` electron. The first major step is understanding the difference between two major theories: Compare the bond order to that seen in the lewis structure (remember that an electron in an antibonding orbital cancels the stabilization due to bonding of. The rules for building up pi molecular orbital; It describes the formation of bonding and antibonding molecular o. Web however, recall that the more electronegative atom will be lower on the diagram. Determine the total number of valence electrons in the he 2 2 + ion. Two for the allyl cation, three for the allyl radical, and four for the. They are usually used to explore domain concepts, understand software requirements and. Web mo diagram for hf. “ the only way to make sense of change is to plunge into it, move with it, and join the dance. Recall that a cation indicates a loss of `1` electron. The mo diagram will be the same as the mo diagram of. They are usually used to explore domain concepts, understand software requirements and. Web toto macau | live draw toto macau | live ttm | live toto macau | live draw ttm | live macau | macau🔴live draw toto macau, livedraw toto macau, live draw tot. Mo diagrams can be built from group orbitals and central atom orbitals by considering orbital. Construct a qualitative molecular orbital diagram for chlorine, cl 2. Web this chemistry video tutorial provides a basic introduction into molecular orbital theory. The mo diagram will be the same as the mo diagram of `o_2`, except with `1` less electron. We need to know what orbitals we are using. Determine point group of molecule (if linear, use d. Try the following mo's on your own, and then check with the answers provided. Web however, recall that the more electronegative atom will be lower on the diagram. The rules for building up pi molecular orbital; N = # electrons in bonding orbitals. The symmetry of group orbitals is determined by reducing a reducible representation of the orbitals in question. In the event of a discrepancy, the official drawing results shall prevail. 2h 2v d ∞h or c ∞v) (z axis is principal axis; Web example 1 1: Bond order gives you an idea of the strength of the bond between two atoms. Web mo diagram for hf the ao energies suggest that the 1s orbital of hydrogen interacts mostly with a 2p orbital of fluorine. Draw the mo diagram for `o_2^+` this is a bit of a curveball, but a perfectly valid problem. This approach is used only when the group orbitals are not obvious by inspection. N* = # electrons in antibonding orbitals. As a rule of thumb, a bond order = 1 equates to a single bond, a bond order = 2 equates to a double bond, etc. Web how to draw molecular orbital diagrams for conjugated systems Valence bond theory and molecular.
how to draw molecular orbital diagrams for polyatomic molecules

MO Diagram Overview, How to draw MO Diagram and Solved example along

Molecular Orbital Diagrams simplified by Megan Lim Medium
How To Draw Mo Diagram Wiring Site Resource

How to Draw Molecular Orbital Diagrams (MO DIAGRAMS) Explanation YouTube
How To Draw A Molecular Orbital Diagram Elevatorunion6

MO Diagram Overview, How to draw MO Diagram and Solved example along
MO Diagrams

Molecular Orbital Diagrams simplified by Megan Lim Medium
MO Diagrams
They Are Usually Used To Explore Domain Concepts, Understand Software Requirements And.
We Are Only Going To Consider Valence Orbitals.
Notice That The Bonding Orbitals Are Bigger On The Oxygen And The Antibonding Orbitals Are Bigger On.
Draw The Mo For O 2:
Related Post: